

ECLI:NL:RBMNE:2020:1466, Rechtbank Midden-Nederland, C/16/446910 / HA ZA 17-746. 1 [eiseres 1] , wonende in [woonplaats 1] , 2.
[eiseres 2], wonende in [woonplaats 2] , 3. Wonende in [woonplaats 3] , 4. Wonende in [woonplaats 4] , 5. Wonende in [woonplaats 5] , 6. Wonende in [woonplaats 6] , 7. Wonende in [woonplaats 7] , 8. Wonende te [woonplaats 8] , 9. Wonende in [woonplaats 9] , 10. Wonende in [woonplaats 10] , 11. Wonende in [woonplaats 11] , 12.
Wonende in [woonplaats 12] , 13. Wonende in [woonplaats 13] , 14. Wonende in [woonplaats 14] , 15. Wonende in [woonplaats 15] , 16. Wonende in [woonplaats 16] , 17. Wonende in [woonplaats 17] , 18. Wonende in [woonplaats 18] , 19. Wonende in [woonplaats 19] , 20. Wonende in [woonplaats 20] , 21. Wonende in [woonplaats 21] , 22. Wonende in [woonplaats 22] , 23. Wonende in [woonplaats 23] , 24. Wonende te [woonplaats 24] , 25. België: Slachtoffers van medische fouten staan vaak in de kou - SIN-NL. Gill v Ethicon S rl (No 5) [2019] FCA 1905. Gill v Ethicon Sàrl & Ors (No 5) [2019] FCA 1905 In accordance with the practice of the Federal Court in some cases of public interest, importance or complexity, the following summary has been prepared to accompany the orders made today.
This summary is intended to assist in understanding the outcome of this proceeding and is not a complete statement of the conclusions reached by the Court. The only authoritative statement of the Court’s reasons is that contained in the published reasons for judgment which will be available on the Court’s website. This is a representative (or class) action brought by three women: Kathryn Gill, Diane Dawson, and Ann Sanders, each of whom suffered complications after surgery involving the transvaginal implantation of synthetic mesh. At the centre of the case are nine medical devices made from knitted polypropylene, a thermoplastic polymer. Stress urinary incontinence is the involuntary leakage of urine. Kathryn Gill was born in 1970. This was a large case. J&J agrees to $117M settlement over pelvic mesh devices. Johnson & Johnson has agreed to a $117 million multistate settlement over allegations it deceptively marketed its pelvic mesh products, which support women's sagging pelvic organs.
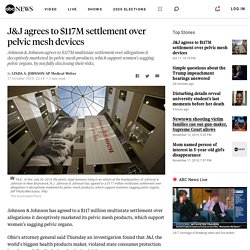
Ohio's attorney general said Thursday an investigation found that J&J, the world's biggest health products maker, violated state consumer protection laws by not fully disclosing the devices' risks. Numerous women who had the once-popular, hammock-like devices implanted claim they caused severe pain, bleeding, infections and other complications. Johnson & Johnson and its Ethicon surgical products unit reached the settlement with 41 states and the District of Columbia. "These companies didn't paint a clear picture of the device's medical risks, preventing patients from making well-informed decisions," Ohio Attorney General Dave Yost said in a statement. Some of the products are still on the U.S. market, and hundreds of thousands of women have had the devices surgically implanted, according to Yost's office.
Verdwaald in een woud van kil papierwerk en procedures. Volkskrant.nl en krant.nl maken onderdeel uit van de Persgroep Nederland.
Onze sites gebruiken cookies en vergelijkbare technologieën (cookies) onder andere om u een optimale gebruikerservaring te bieden. Morrisey sues Johnson & Johnson over marketing of surgical mesh. CHARLESTON – West Virginia Attorney General Patrick Morrisey's office has filed suit against Johnson & Johnson and subsidiaries alleging the companies engaged in unlawful, unfair and deceptive marketing practices associated with their surgical mesh devices.
“Misrepresenting any product’s safety is unlawful and can have dire effects on consumers,” Morrisey said in a Sept. 18 press conference at the state Capitol. “When medical products are marketed in an improper manner, it can put consumers’ health at risk.” The lawsuit, filed Sept. 18 in Monongalia Circuit Court, claims Johnson & Johnson and Ethicon misrepresented the effectiveness, properties, risks and safety history of the surgical mesh products in marketing and educational materials provided to patients and doctors, in personal meetings and in published medical articles.
State attorneys argue J&J missed adequate label warnings in pelvic mesh trial. Hinoul SAN DIEGO – In a trial to decide if Johnson & Johnson sold pelvic mesh implants allegedly knowing they could harm women, state attorneys on Wednesday attempted to get a top company official to admit that risk warnings on the products had been lacking.
Jinsook Ohta, the attorney with the state's attorney general's office, asked if the words “life-long erosion” and possible “re-occurrence” were cited in the warnings of potential risk. “Re-occurrence and life-long (complications) are not cited,” said Dr. Ethicon complaint reviewer says in J&J trial that patients felt vaginal mesh instructions fell short of explaining risks. Chen SAN DIEGO – In a trial to determine if Johnson & Johnson officials sold vaginal mesh implant kits to doctors to make profits knowing the devices were harming women, a complaint reviewer on Wednesday said some sling patients had felt misled about the risks.
“I saw there were (complaint) patterns,” said Dr. Meng Chen, a San Francisco doctor and former medical director for surveillance safety at Ethicon, a J&J subsidiary that made the vaginal mesh that is the subject of the suit. The trial in the San Diego Superior Court is being streamed live courtesy of Courtroom View Network. Pathologist explains how mesh distorts, twists in J&J pelvic mesh trial. Kubota SAN DIEGO – In a trial to determine if Johnson & Johnson injured women by selling pelvic mesh devices the company allegedly knew were dangerous for profit, a pathologist on Thursday said the mesh can distort once it’s implanted and such devices can also migrate in the body.
“Polypropylene is not inert; the mesh is migrating, moving through the tissue and can become exposed,” said Dr. Vladimir Iakovlev, a Toronto-based researcher with St. Michael’s Hospital and an expert witness for the state. “The nerves around the mesh can be affected by it and this will be felt as pain. Second Wave of Transvaginal Mesh Litigation Focused on Serious Injuries. $80 Million Verdict in Ethicon Prolift Pelvic Mesh Lawsuit. The need for transparency of clinical evidence for medical devices in Europe. Privacy settings. Law360. Transvaginal & Hernia Mesh Lawsuit Settlements - Cases and Payouts.
Video: What's a Vagina Worth? Robin's Story. Privacy settings. Deze website maakt net zoals de meeste websites gebruik van cookies.
Via deze cookies verzamelen wij informatie over uw bezoek en interesses. Op deze manier is het mogelijk om onze inhoud blijvend gratis aan te bieden en een gepersonaliseerde website en relevante advertenties te tonen. Zo houden we het voor u op elk moment interessant. Patients with faulty Johnson & Johnson hip implants to be compensated up to Rs 1.23 crore. NEW DELHI: The health ministry on Thursday approved an expert committee's proposed formula to compensate patients who had received faulty hip implants made by a subsidiary of US medical device giant Johnson & Johnson.
The approved formula would see patients being compensated anywhere between Rs30 lakh and Rs1.23 crore, depending on the age and level of disability of the patient, according to a release by the ministry. The formula include a base compensation of Rs 20 lakh and an additional Rs 10 lakh for 'non pecuniary' damages, which are damages that cannot be readily quantified. "This will be a big relief for patients," health secretary Preeti Sudan told ET. "This has been done in a consultative process," she added. J&J globally recalled these implants in 2010, including from India, after a British study found the models to have higher failure rates, which means there was a higher chance of patients requiring a revision surgery to replace them.
Flash - 400 Swedish women win compensation over faulty French breast implants - France 24. A French court on Thursdayordered the German safety certifier that approved defective breast implants for use to compensate around 400 Swedish women who received the devices behind a global health scare. The court in the Paris suburb of Nanterre ordered TUV to pay 4,600 euros ($5,200) to each of the Swedish plaintiffs for the harm caused by approving implants made by French firm Poly Implant Prothese (PIP). It also ordered the company to reimburse the cost of having the implants -- which were filled with a cheap industrial-grade silicone gel used in electronic equipment -- removed.
It comes a month after France's highest appeals court ordered that TUV be retried for negligence over its role in the affair, after being initially cleared of liability by a lower court. After a separate trial the company had already been ordered to pay 60 million euros ($69 million) as a deposit for potential compensation for 20,000 women who received the defective implants. Edwards et al v. Ethicon, Inc. et al, No. 2:2012cv09972 - Document 139 (S.D.W. Va. 2014)
Court Description: MEMORANDUM OPINION & ORDER (Daubert Motions) for the reasons set forth herein, denying 73 MOTION by Ethicon, Inc., Johnson & Johnson to Exclude the Opinion Testimony of John F. Steege, M.D.; denying 97 MOTION by Ethicon, Inc., Johnson & Johnson t o Exclude the Opinions and Testimony of Dr.
Scott Guelcher, Ph.D.; denying in part, denying as moot in part and granting in part 75 MOTION by Ethicon, Inc., Johnson & Johnson to Limit the Testimony of Bruce Rosenzweig, M.D.; denying in part, denyin g as moot in part and granting in part 85 MOTION by Ethicon, Inc., Johnson & Johnson to Exclude Testimony of Vladimir Iakovlev, M.D.; denying in part, granting in part and reserving in part 77 MOTION by Ethicon, Inc., Johnson & Johnson to Exclude Certain Opinions of Jerry G. Privacy settings. Deze website maakt net zoals de meeste websites gebruik van cookies.
Via deze cookies verzamelen wij informatie over uw bezoek en interesses. Fonds Medische Ongevallen schakelt Zwarte Lijst verzekeringsarts Marc De Kock in om dossiers slachtoffers medische fouten af te wijzen. Unneeded Transvaginal Mesh Implants Removed for Profit - Saunders & Walker PA. In recent years, there have been a growing number of transvaginal mesh lawsuits brought against the manufacturers like Johnson & Johnson, Ethicon, Bard, Boston Scientific, Coloplast, and Cook.
In some situations, after a pelvic mesh attorney files a claim against the manufacturer, the case is then combined into one of several vaginal mesh class action lawsuits. The problem with the transvaginal mesh was it was not fully tested before being released on the market. The FDA (Food & Drug Administration) mistakenly fast-tracked the approval process based on previous approvals for other surgical mesh products like the mesh used to repair hernias.
The manufacturers of transvaginal mesh products took advantage of this FDA’s 501(k) program—the program that allowed them to sidestep the normal approval processes. In addition, the FDA did not even take the time to require any type of studies to determine any potential side-effects or risks of using the transvaginal mesh products. Ethicon Mesh Lawsuit Alleges Prolene Hernia Patch Entangled Man's Spermatic Cord - AboutLawsuits.com. Can Transvaginal Mesh Surgery Be Reversed? Thompsons Solicitors welcomes senior UK doctors’ calls for public inquiry into vaginal mesh surgery. Thompsons Solicitors’ clinical negligence team has welcomed calls for a public inquiry into vaginal mesh surgery, following concerns that complication rates may be higher than official figures imply. Tens of thousands of women in England are believed to have been treated with vaginal mesh implants, slings and tapes in the past decade, according to figures obtained by the Guardian.
The surgery sees a plastic mesh inserted to support the bowel, bladder or womb, often to treat childbirth complications, such as urinary incontinence and organ prolapse. However, at a meeting in parliament organised by Pontypridd Labour MP Owen Smith, campaign group Sling the Mesh called for the practice to be stopped. Carl Heneghan, professor of evidence-based medicine at the University of Oxford, and Sohier Elneil, consultant urogynaecologist at University College Hospital, said complication rates for some types of the surgery appeared to be unacceptably high. You can also read our TVT factsheet here. Johnson & Johnson Pelvic Mesh Defensive Win Reversed - Arentz Law Group. By Emily Cox An Ohio woman has won a damages hearing a little more than a month after a jury found that Johnson & Johnson pelvic mesh was defective but did not cause her injuries.
Philadelphia Court of Common Pleas Judge Michael Eros granted plaintiff Kimberly Adkins’ post-trial motion this past week. On June 9, a Philadelphia jury sided with manufacturers in the Johnson & Johnson pelvic mesh trial despite defensive lawyers admitting during closing arguments that there was troubling evidence that the companies had rushed TVT Secur to market despite substantial internal concerns over the product’s safety. In particular, the device showed the potential for high failure rates and medical complications. The jury conceded that the device was defective. The first case to go to trial out of the mass tort program yielded a $12.5 million verdict in December 2015.
America’s Lawyer [21]: Facebook Murders, Lead in Los Angeles & Dangerous Mesh. Transvaginal Mesh Recall - Discontinued & Recalled Mesh. Transvaginal mesh was first approved in the 1990s by the Food and Drug Administration (FDA) for the repair of pelvic organ prolapse and stress urinary incontinence in women. It is a mesh like medical device that is constructed out of plastic or biological material, most commonly a plastic coated with biological materials. Transvaginal Mesh Global Roundup: Sensational, Scandal and Settlement. British tabloids have sensationalised transvaginal mesh by describing one victim’s botched surgery a “biting vagina”; an Australian Senator compares the mesh to Thalidomide as one of Australia's worst health scandals; and in the US a transvaginal mesh class action lawsuit is settled for $12.3 million.
Sensationalized in the UK: The Biting Vagina The “Biting Vagina” story first told by Theresa Bartram on her Facebook page has been sensationalized by the Daily Mail, reporting that she “was left with a Venus flytrap vagina that 'bit' her partner during sex after surgery for incontinence that went horribly wrong.” Bartram’s TVT led to serious problems including ‘ripping a chunk’ from her partner’s penis. Trust a British tabloid to pen this: “It was like [my vagina] had grown teeth,' she recalled. His [penis] was bright red and spouting blood. Bartram is now campaigning for the procedure to be banned in England. Dr. IN RE ETHICON, INC., PELVIC REPAIR SYSTEM PRODUCTS LIABILITY LITIGATION. Transvaginal Mesh Deaths Reported as Injuries? “A good number of attorneys have reported transvaginal mesh adverse events to the FDA on behalf of their clients/patients, but over 23,000 reports are from physicians,” says Madris Tomes, founder of Device Events and former FDA Program Manager.
“Physicians are not great at reporting— this is a HUGE number.” During her tenure at the FDA, Tomes worked on the agency’s adverse event reporting program. She can search the entire FDA medical device adverse events data since 1996. For instance, Tomes recently investigated Essure complaints and found 303 reports referencing these fetal “death like” terms—reported as injuries rather than deaths-- and provided the information to Congressman Mike Fitzpatrick with the goal to remove Essure from the market.
Turning her attention to transvaginal mesh, Tomes looked at the FDA’s MAUDE database that houses reports of adverse events involving medical devices. The FDA data also shows 126,349 reports of mesh adverse events and 49 recalls. Even More Warnings for Transvaginal Mesh - Dangerous Medicines. Transvaginal Mesh Complications May Take Risky Product Off Market. The FDA has recently issued a press release announcing that the makers of surgical mesh products used to treat pelvic organ prolapse (POP) must reclassify the devices as Class III, or high risk medical devices.
Pelvic Mesh Likely to See Piecemeal, Not Global, Resolution. Transvaginal Mesh: Chilling Allegations in State Lawsuits Against Johnson & Johnson — North Carolina Product Liability Lawyer Blog. American Medical Systems Settles over 100 Pelvic Mesh Suits - Legal Reader. AGs: Pelvic mesh implants caused dire complications.
According to California's attorney general, J&J sold more than 787,000 pelvic mesh devices in the United States from 2008 until 2014, including more than 42,000 in California. The lawsuits, which were filed in California and Washington state, allege that Johnson & Johnson failed to disclose a host of problems caused by the devices, which are created to treat the conditions stress urinary incontinence and pelvic organ prolapse. Transvaginal Mesh Nerve Damage “Horrendous” Vaginal Mesh Lawsuit Upheld by Appellate Court. To Webcast $6M+ Boston Scientific Pelvic Mesh Trial In Massachusetts.
Why The Transvaginal Mesh Litigation Won't Become A "Black Hole" Like Asbestos. Allergic to AMS Transvaginal Mesh. Video.wttw. Urogynecology expert witness says manufacturer failed to disclose problems with transvaginal mesh. Transvaginal Mesh—The Past Five Years. BLADDER SLING ATTORNEYS. Reckless: American attorney savages multinational healthcare manufacturer over faulty mesh implant used in Scotland. Johnson & Johnson loses another pelvic mesh suit. The Legal Intelligencer. When a Brain Surgeon Becomes a Malpractice Lawyer. Verdicts in Philadelphia Pelvic Mesh Cases Troubling for Johnson & Johnson - NewsInfernoNewsInferno.
AMS Transvaginal Mesh Settlement Money “Chump Change” Cases filed in the West Virginia Southern District Court Before Judge Joseph Goodwin. Johnson and Johnson Hit with 13.5M Pelvic Mesh Verdict - Legal Reader. Boston Scientific, C.R. Bard Beat $28M Pelvic Mesh Lawsuit - Legal Reader. Mostyn Law wins court order barring Boston Scientific from destroying evidence. Safety Concerns for Patients of Gynaecologist Dr Angamuthu Arunkalaivanan - Bolt Burdon Kemp.
Delay damages added to verdict in vaginal mesh case. Victims of Transvaginal Mesh Devices Finally Get the Justice they Deserve. UK concerned that reforms to medical device regulations could increase NHS costs. Pelvic mesh MDL ‘most complicated MDL in history,’ plaintiff attorney says. Valeant Pharmaceuticals CEO: We’re in Business of Shareholder Profit, not Helping the Sick. Recent TVM settlements, verdicts may lead to resolution in outstanding MDL. Trans Vaginal Mesh Opt-Outs Continue, but there is a Serious Problem with... First Do No Harm: Complications and Complaints will Change Pelvic Floor Surgery Using Mesh Implants in 2015. Transvaginal Mesh Lawsuit News: Another Joint Conference Status Meeting Ordered by Judge to Resolve More than 70K TVM Lawsuits against Several Mesh Manufacturers, Says Lawsuitsettleme... ( CHARLESTON W.Va. April 27 2015 /P...) Team 10: Woman says caller asked her to lie about controversial surgery.
Papantonio: Johnson & Johnson Lies Destroyed Women's Lives. Viewpoint: Transvaginal mesh jury award should spur recall, banDelaware Business Daily digital business journal. No retrial for Ethicon in $3m pelvic mesh loss – MassDevice. Judge Consolidates 26 Ethicon Vaginal Mesh Lawsuits Into One November Trial. Ex 10.144 Mesh Settlement Agreement. Lawsuits being settled in surgical mesh complications. # Van der Goen Advocaten. Report: Bard to pay $200m to settle mesh cases – MassDevice. Class actions aren’t the only game in town for product liability claims.
Ill. Woman Joins Transvaginal Mesh MDL. Transvaginal Mesh - Alamo Journal. Judge Won’t Dismiss Vaginal Mesh Punitive Damages Request. COOK MEDICAL PREPARES FOR TRIAL OVER ITS PELVIC MESH PRODUCTS. AMS Transvaginal Mesh Complaint: Is My Sex Life Over? Papantonio: Faulty Mesh Maims Thousands of Women. Consolidated Trials for Ethicon Mesh Lawsuits Sought Given Lack of Settlement Progress.
Lawsuits being settled in surgical mesh complications. Coloplast Will Pay $16 Million in Vaginal Mesh Settlements. *New* Mesh Maker Covidien Agrees to Settle 11,000 Claims. Vaginal Mesh Lawsuits Inching Towards Settlement. Lawsuit Help Center Reports $100MM Verdict against Boston Scientific. Medical Case Reviews for Women Injured by Trans Vaginal Mesh to Help Determine if "Opting Out" is Best.
How Law Firms Use Facebook and Other Data to Track Down Medical Victims. Covidien agrees to settle some cases over transvaginal mesh devices. FinalMasterComplaint.pdf. Why Did Ethicon Withdraw Mesh From the Market? - Florida Mass Tort Attorneys, Lawyers. $100 Million verdict against transvaginal mesh implant manufacturer. Transvaginal Mesh Death: Beyond Statute of Limitations? Recent Transvaginal Mesh Lawsuits Could Affect Georgia. Blogger urges others injured by surgical mesh to file report.