

Breastfeeding and the Gut Microbiome. Necrotizing Enterocolitis. Should You Feed Your Infant Gluten? Infant Gut Microbiome Facts - Infographic. How Infants Acquire Their Gut Flora.
This is a post from the Gut Critters blog that ended November 18, 2016. Ray Medina gave permission for his material to be copied as long as it was attributed to him and not used for commercial purposes. – kiraonysko
Video: The assembly of an infant gut microbiome framed against healthy human adults. Infant mycobiome varies by body site and mode of birth during the first month of life. Gut’s Earliest Bacterial Colonizers. Infants start out mostly microbe-free but quickly acquire gut bacteria, which take root in three successive groups.

First, Bacilli dominate. Then Gammaproteobacteria surge, followed by Clostridia. But the pace at which these bacterial groups colonize the gastrointestinal tract depends on the time since the babies were conceived, not since when they were born. And time since conception appears to have more of an influence on the infant gut microbiome than other factors, such as exposure to antibiotics, whether babies were born vaginally or by cesarean section, and if they were breastfed.
These are a few of the findings from a survey of 922 fecal samples collected from 58 premature babies, published today (August 11) in PNAS. “I think the paper does a nice job of showing that premature babies develop differently from full-term babies . . . it is not just a function of colonization after birth,” Rob Knight from the University of Colorado, Boulder, told The Scientist in an e-mail.
The infant gut microbiome: New studies on its origins and how it's knocked out of balance. A fecal sample analysis of 98 Swedish infants over the first year of life found a connection between the development of a child's gut microbiome and the way he or she is delivered.

Babies born via C-section had gut bacteria that showed significantly less resemblance to their mothers compared to those that were delivered vaginally. Gut Flora In Infants and Children. Bifidobacteria colonization in newborns. Bifidobacteria is a key marker of a healthy gut flora in infants.
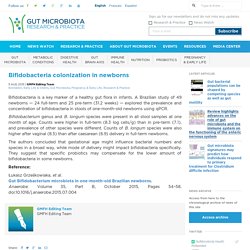
A Brazilian study of 49 newborns — 24 full-term and 25 pre-term (31.2 weeks) — explored the prevalence and concentration of bifidobacteria in stools of one-month-old newborns using qPCR. Bifidobacterium genus and B. longum species were present in all stool samples at one month of age. Counts were higher in full-term (8.3 log cells/g) than in pre-term (7.1), and prevalence of other species were different.
Counts of B. longum species were also higher after vaginal (8.5) than after caesarean (6.9) delivery in full-term newborns. The authors concluded that gestational age might influence bacterial numbers and species in a broad way, while mode of delivery might impact bifidobacteria specifically. Pacifier Cleaning and the Risk of Allergy Development. Cesarean Section, Formula Feeding, and Infant Antibiotic Exposure: Separate and Combined Impacts on Gut Microbial Changes in Later Infancy.
Introduction The human gut microbiome is assembled throughout infancy with an age-specific succession of microbes after birth.
This assembly is characterized by increasing colonization with bifidobacteria and microbes belonging to the Firmicutes and Bacteroidetes phyla, and depletion of the first colonizers, Enterobacteria (1–7). With the introduction of solids, enrichment is also seen with the beta-Proteobacteria. Cesarean delivery and maternal intrapartum antibiotic prophylaxis delay this trajectory of microbial deposition, while formula feeding can modify it (1, 3). Quality matters in probiotic products - Gut Microbiota for Health. Bacterial exposures necessary for decreasing risk of allergy. Observational studies in humans have shown that early life exposure to microbes in a variety of situations is associated with a decreased risk of asthma.
These include: – Exposure of a pregnant mother to microbes in order to protect her baby – Exposure of children to livestock or dogs – Growing up with numerous siblings Meanwhile, disruption of normal colonization processes (such as antibiotic use and C-section delivery) is associated with an increased risk of allergy. Some gut microbiota changes also track with asthma risk. For example, one study showed a greater risk of developing childhood allergic diseases when infant feces contained higher levels of Escherichia coli or Clostridium difficile. Whereas observational studies don’t tell us about causality, mouse models can be useful first steps for identifying causal links. A recent mouse study delved more deeply into how microbial exposures were associated with allergic immune responses. Newborn Immune Systems Suppressed. WIKIMEDIA, ERNEST FFrom the sterile world of the womb, at birth babies are thrust into an environment full of bacteria, viruses, and parasites.

They are very vulnerable to these infections for their first months of life—a trait that has long been blamed on their immature immune systems. But Shokrollah Elahi from the Cincinnati Children’s Hospital Medical Center has shown that, at least in mice, this susceptibility is the work of special cells that actively suppress immune responses in newborns. This raises their risk of diseases, but it also creates a window during which helpful bacteria can colonize their guts. The results are published today (November 6) in Nature. “This more intricate regulation of immune responses makes more sense than immaturity,” said Sing Sing Way, who led the study, “because it allows a protective response to be mounted if needed.” The team began by injecting 6-day-old mice with splenocytes (a type of white blood cell) from adults. S. The maturation of the microbiome during the first year of life — The American Microbiome Institute.
The team of scientists studied 98 women and their newborn babies.

They sequenced the mother’s stool, the newborns stool, and again the child’s stool at 4 and 12 months. Throughout the study, because they used a technique called shotgun sequencing, they identified 4,000 new microbial genomes. The infants in the study were breastfed for varying amounts of time with some never being breastfed at all. The researchers found that breastfeeding and the timeline of cessation of breastfeeding was critical to driving microbiome development. Many had previously hypothesized that it was the time at which solid foods were introduced was most important for microbiome development, however this study found that it was the time at which breastfeeding was stopped. Exposure to household furry pets influences the gut microbiota of infants at 3â4 months following various birth scenarios. In this general population cohort of 746 Canadian infants, we observed higher overall species richness and changes to taxon abundance in gut microbiota of infants exposed to furry pets during pregnancy or continuing to the postnatal period.
These findings are in agreement with our previous report [17] of postnatal pet exposure on 3-month gut microbiota but not with studies in later infancy [26]. Moreover, elevations in microbial species richness in this study were evident with prenatal pet exposure. Since several studies including our own within the same cohort have reported low microbiota richness in early life to be associated with food sensitization and other atopic diseases [20, 27, 28], higher microbial richness with prenatal pet exposure may confer protection against the development of atopy. Early-Life Microbiome. Analyzing the gut microbiomes of children from birth through toddlerhood, researchers tie compositional changes to birth mode, infant diet, and antibiotic therapy.

V. ALTOUNIAN/SCIENCE TRANSLATIONAL MEDICINEAnalyzing stool samples from 39 children in Finland for the first three years of their lives, a team led by investigators at the Broad Institute monitored the effects of factors including birth mode (vaginal versus Cesarean-section delivery) and the administration of antibiotics. Early-In-Life Probiotics Reduce Risk of Diabetes, Otitis Risk. Mucin Cross-feeding Shapes The Metabolic Environment In The Infant Gut. The infant microbiome changes before the onset of type 1 diabetes — The American Microbiome Institute.
The researchers sampled the stools of 33 infants in Finland and Estonia that were genetically at-risk for diabetes.

Their first major discovery was that even though the bacterial composition of the microbiome grew, changed, and became more diverse with age, the types and number of genetic pathways that were expressed by the microbiome, as well as the metabolites produced by the microbiome remained stable. They also found many similar bacterial species between infants, however these infants usually had different strains of said bacterial species. In most of these cases, once a particular strain established itself in the gut it remained stable and would not be displaced.
The scientists tracked the microbiome changes that occurred with diet as well. During breast feeding Bifidobacterium and lactobacillus predominated, and Lachnospiraceae decreased. These results provide exhaustive evidence for an association between the microbiome and diabetes. New research shows that Bifidobacteria transfer from mother to child — The American Microbiome Institute. Both natural birth (as opposed to birth by C-section) and breastfeeding are topics that stir up a lot of conversation among mothers and the scientific community. For example, there is the question of whether breastfeeding rather than formula feeding has some specific benefit to an infant’s health. Well, what about the infant’s gut microbial health? A new article published by Applied and Environmental Microbiology takes a look at whether natural birth and breastfeeding coincides with an exchange of bacteria from mother to child. Mother’s Microbes Protect Baby’s Brain.
V. ALTOUNIAN/SCIENCE TRANSLATIONAL MEDICINEThe brain—the most exalted and enigmatic of organs, which is closed off from the rest of the body by a largely impermeable barrier—could not seem more disconnected from the intestine. Yet, according to a paper published today (November 19) in Science Translational Medicine, it’s thanks to the contents of the gut—specifically, the resident bacteria—that the mouse brain’s impermeable barrier develops properly, both before and after birth. “It’s absolutely fascinating to think that gut bacteria can control permeability of the blood-brain barrier,” said Caltech microbiologist Sarkis Mazmanian who was not involved in the study. “Many neuroscientists staunchly believe that the blood-brain barrier is an incredibly impermeable membrane to many molecules and . . . would hardly believe that gut bacteria would control such an integral part of our neurobiology.”
It’s also known, he said, “that [gut] microbes could modulate brain function and development.” Early life stress implications on the gut microbiome — The American Microbiome Institute. A growing body of evidence supports the significance of the gut microbiome with respect to behavioral disorders, as mediated by disruption to the gut-brain axis. Importantly, there is a lack of understanding regarding associations between gut microbiome dysbiosis and behavioral phenotypic outcome. Traumatic childhood events early-in-life can result in later-in-life behavioral consequences. Maternal separation (MS) is an example of such an event that is represented with a well-established preclinical (i.e., animals) experimental model for early life stress. Maternal stress can alter the gut microbiome of progeny, possibly affecting brain development — The American Microbiome Institute.
The Maternal Microbiome. WIKIMEDIA, JEAN HOUSENPopular thinking has held that as a baby works his way through a birth canal teeming with microorganisms, his body is colonized with its first commensal bacteria. But a new study shows that a bevy of microbes exist in the womb. The findings, published today (May 21) in Science Translational Medicine, add to a growing body of literature suggesting that tissues once thought to be germ-free are crawling with microbes, and that babies’ introduction to the microbial world comes from multiple maternal sources. “[It’s an] interesting study that continues to build the snowball that no tissue in the human body is sterile, including reproductive tissues and, for that matter, the unborn child,” Seth Bordenstein, a biologist at Vanderbilt University who was not involved in the work, said in an e-mail to The Scientist.
Hints that the uterine environment harbors bacteria began to emerge several years ago. What is a Baby’s Microbiome and Why Should Expectant Mothers Care? Microorganisms in the womb set stage for diseases. How probiotics can help ward off health complications in premature babies. Each year, about 29,000 babies are born prematurely in Canada. Premature infants are at an increased risk of infections and even death, but probiotics are increasingly being used as a safe and effective way to prevent health complications.
A year ago, the neonatal intensive care unit at BC Women’s Hospital in Vancouver started giving babies born prematurely and weighing 1,500 grams or less five strains of so-called good bacteria on a daily basis. Randomized clinical trial shows probiotic may not be an effective treatment for colic — The American Microbiome Institute. Many families have experienced colicky infants who have excessive and inconsolable crying. The cause of this behavior is largely unknown, however it is beginning to be linked to a variety of diseases, including allergies and gastrointestinal disorders. Antibiotics decrease microbiome diversity in premature infants — The American Microbiome Institute. Exactly What We Expected: “Bacterial Signature” Found in Colicky Babies.
A few days ago, I noted that the association between smoking and baby colic was another piece of evidence pointing to a broader smoking/microbiota link. Gut microbiota and infant vaccine protocol. Why do some children suffer adverse vaccine events while others escape injury? What can be done to limit risk of injury by vaccination? Current CDC (Centers for Disease Control and Prevention) vaccine protocol beginning within 12 hours of birth does not factor gut microbiota as crucial to immune response.
Maternal HIV infection can disrupt the gut microbiota development of infants - Gut Microbiota for Health. Worldwide each year more than a million infants are born to women infected with human immunodeficiency virus (HIV), the virus causing AIDS. Microbiome disruptions could explain HIV-exposed babies’ increased risk of morbidity and mortality - Gut Microbiota for Health. It is already known that a reduction in gut microbial richness is the hallmark change of human immunodeficiency virus (HIV) infection, but how this dysbiosis is established in the HIV-exposed uninfected infant is poorly understood.
A population based study of S. aureus colonization in infants for atopic eczema — The American Microbiome Institute. Atopic eczema (AE) is a skin condition that is often measured by transepidermal water loss (TEWL), a mark of dry skin. The ailment is often associated with the colonization of Staphylococcus aureus and dysfunction of the skin barrier (a few months ago we had a long blog post about atopic eczema and S. aureus so if you're interested in this topic, take a look at that post as well). Infant Gut Microbiome Associated With Cognitive Development. Periodontitis and its possible contribution to preterm birth — The American Microbiome Institute. Evidence is accumulating that gum disease can lead to chronic systemic inflammation throughout the entire body.
The oral microbiome of children, and its relation to dental caries — The American Microbiome Institute. The microbiome may affect a child’s temperament — The American Microbiome Institute. Gut bacteria may help prevent asthma in children — The American Microbiome Institute. Intestinal Microbiota Composition in Sudden Infant Death Syndrome and Age-Matched Controls. The nasal microbiome of infants may impact risk of developing asthma — The American Microbiome Institute. Four types of gut bacteria to protect you of asthma. Are clues about childhood asthma and heightened immune responses found in a baby’s gut microbiome? - Gut Microbiota for Health.
Finding early-life microbiota markers of asthma risk. The microbiome of children with short bowel syndrome — The American Microbiome Institute. The Human Microbiome and Its Potential Importance in Pediatrics.