

Early COVID-19 Vaccines 'Really Encouraging,' Says NIH Boss. It isn’t often that one hears the word “pandemic” and “inspired” in the same sentence.
But the director of the U.S. National Institutes of Health, Dr. Francis Collins, says that, for all the suffering and grief that COVID-19 has wrought around the world, he has at least witnessed an unprecedented level of cooperation between private industry and the alphabet soup of government agencies who are urgently seeking a vaccine for the devastating virus. “We’ve never had that before. The Oxford Vaccine: Innovation for the Global Good. AstraZeneca vaccine scientists set for £22m payday in New York float. Prof Sarah Gilbert, the scientist who led the team that created the Oxford/AstraZeneca coronavirus vaccine, is set for a payday of more than £20m as the biotech firm she co-founded prepares to float on the stock market in the US.
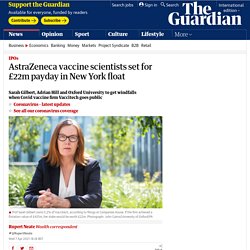
Gilbert, who became a household name as a result of her work creating Oxford’s Covid-19 vaccine, owns 5.2% of Vaccitech, a Oxford University spin-out company that owns the biotechnology behind the AstraZeneca vaccine and others for Mers, hepatitis B, the virus that causes shingles, and a range of cancers. The company, which Gilbert founded in 2016 with a fellow Oxford vaccine scientist Prof Adrian Hill, was reported on Wednesday to have filed regulatory papers before an initial public offering on the Nasdaq exchange. The report in the Financial Times did not suggest a guideline valuation for Vassitech, but it is likely that the company will seek a flotation price in excess of the £425m valuation it attracted in a fundraising drive earlier this month. Safe, stable, sold at cost: AstraZeneca’s vaccine deserves celebration, not scorn.
AstraZeneca is one of the shining stars of the pandemic.
Not only did it produce a vaccine where other big players failed, the UK-Swedish company has pledged to sell it at cost until it is able to declare the pandemic over. Because the vials that contain Astra’s vaccine can be kept in a normal refrigerator, it has managed to keep the cost down to about $3 (£2.20) a shot, compared with the $35 charged by US firm Moderna for its vaccine outside the States. The low cost is one major benefit, but it is matched by the ease of transporting the vaccine, which puts it at the top of the list for use in developing countries. As a result, there is almost limitless demand for the vaccine from across the globe. Astra has been attempting to ramp up production rapidly, but has hit problems. Draft landscape and tracker of COVID-19 candidate vaccines.
Overview The COVID-19 candidate vaccine landscape and tracker database compiles detailed information on COVID-19 vaccine candidates in development.
The landscape is updated regularly - twice a week (Tuesday and Friday, 17:00 CET). The landscape: Vietnam’s in-house vaccines will play a role in pandemic geopolitics. Vietnam has pushed domestic vaccine development in part to reduce reliance on any single foreign source.
But its willingness to share vaccine technology and support production abroad may help raise the country’s geopolitical status among the developing world. Editorial. Did Russia just develop a fully working COVID-19 vaccine? The Lancet study, explained. Almost a month after Russia announced approval for 'Sputnik V' COVID-19 vaccine, the first peer-reviewed studies published in the medical journal The Lancet noted that the vaccine evoked antibody response in all participants with no serious adverse events in small human trials.
The Russian coronavirus vaccine, from the day it was first announced to huge fanfare, has split opinion. While experts have questioned the rapidness with which the vaccine tests were done, and the lack of transparency in the results, Russian president Vladimir Putin had assured that it had passed the required checks, adding that one of his daughters was administered the vaccine. Russia's Sputnik V Covid vaccine gaining acceptance in Europe. Russia’s Sputnik V vaccine appears to be gaining acceptance in the European Union, as the head of Germany’s regulatory authority publicly praised the Covid-19 jab and Italy could become the first European country to produce the vaccine from the summer.
Thomas Mertens, the head of Germany’s standing commission on vaccination, described Sputnik V in an interview on Wednesday as “a good vaccine that will presumably also be approved in the EU at some point”. “Russian scientists are very experienced in vaccines,” Mertens told Rheinische Post. “Sputnik V is a very clever construct.” Russia drew criticism last summer when it approved Sputnik V for widespread use after less than two months of human testing, amid fears that the Kremlin was hoping to leverage the vaccine as a soft-power tool.
: What do we know about China's coronavirus vaccines? Oxford vaccine: How did they make it so quickly? The Politics Behind China’s COVID-19 Vaccine Choices. With two new COVID-19 vaccines having just been given the green light to roll out in China, four distinct brands are now available to the Chinese population, and all four are made in China.
One might think that China’s choice to use, for the moment at least, only domestically-produced COVID-19 vaccines is a function of market forces, particularly supply and demand, and cost. And if Chinese vaccines were achieving more or less the same efficacy in trials, and effectiveness among the vaccinated population as some vaccines made outside of China are reaching, then it would make sense that Beijing would rely primarily upon its own domestic pharmaceutical manufacturing capabilities to provide for its own population. If on top of that production came in at a likely lower cost than foreign vaccines, then the business model would be perfectly logical. But that is not the case.
Daxue says that China currently holds 11 percent of the global pharmaceutical market. How Pfizer’s Head Of Vaccine Development Made Covid-19 Vaccine In Record Time And Led A Large Team. Few people have led large-scale scientific research projects, and even fewer have spearheaded development of life-changing vaccines.
Kathrin Jansen is one of those few who has made a mark in science and global healthcare. She initiated and led development of the world’s first vaccine for cervical cancer, Gardasil, and most recently, in her role at Pfizer, helped orchestrate the record-time creation of a Covid-19 vaccine. Johnson & Johnson Vaccine Scientist Exclusive Interview. Meet a Janssen Scientist Developing a Potential Novel Coronavirus Vaccine. Eeks before many people had even heard of the novel coronavirus, Roland Zahn, Ph.D., was already studying the disease—specifically, how it could be stopped.
Share Did you like reading this story? Click the heart to show your love. expand Roland Zahn, Ph.D., Senior Scientific Director of Preclinical Immunology of Viral Vaccines, Janssen. Explained: What is Covaxin, India’s Covid-19 vaccine candidate; how long before approval? Covaxin coronavirus vaccine candidate: Bharat Biotech plans to begin its phase I and II trials in July, but is unsure of the overall timeline for testing and approving its vaccine. The Central Drugs Standard Control Organisation (CDSCO) has allowed Bharat Biotech India (BBIL) to conduct human clinical trials for ‘Covaxin’, an indigenously developed Covid-19 vaccine candidate.
Trials are scheduled to start across India in July. What is ‘Covaxin’ and how was it developed? Yahoo fait désormais partie de Verizon Media. Bharat Biotech's COVID-19 shot 81% effective, Indian firm's interim data shows. India at heart of global efforts to produce Covid vaccine. As the largest global supplier of drugs and producer of 60% of the world’s vaccines, India has long been known as the “pharmacy of the world”. Now, as the frenzied hunt for a Covid-19 vaccine gathers momentum, the country is playing an increasingly strategic and central role in the development, manufacturing – and, crucially, possible future distribution – of several possible Covid shots. With more than 7.5 million cases and about 115,000 deaths, India is also one of the worst affected countries by the virus, second only to the US. A deal has already been struck for the Serum Institute of India, based in the city of Pune, to produce 1bn doses of the the Oxford/AstraZeneca vaccine, seen as the forerunner in the vaccine race.
In anticipation of its success, it has already begun production of almost 2m samples of the vaccine and is carrying out phase 3 human clinical trials on thousands of patients spread across 15 Covid-19 hotspots in India. COVID-19: The inside story of the fastest vaccine ever developed. Jennifer Haller layered a blue denim shirt over a gray tank top that morning, knowing she would later need to bare one shoulder.
Her husband, always on her case about skipping breakfast, scrambled some eggs. The night before, she had read news online that the first person would get an experimental vaccine against the novel coronavirus at Kaiser Permanente in the morning. She wondered if that person might be her. When she arrived at the research center, not far from Seattle’s iconic Space Needle, the whole world was standing by. It had been 75 days since news broke of unexplained cases of pneumonia seeming to emanate from a seafood market in Wuhan, China, and 66 days since scientists in the United States stared at the virus’s genetic code and vowed to conjure a vaccine to shut it down, at a record-shattering pace.