

Mesh-injured women demand review of every removal amid fears that scores of life-threatening implants remain in place despite surgery. Claim of 'professional conspiracy' against US mesh surgeon. A US vaginal mesh surgeon cancelled his trip to Scotland to help women affected by the surgery due to a "professional conspiracy" against him, it has been claimed.

Scottish Conservatives interim leader Jackson Carlaw demanded answers from First Minister Nicola Sturgeon after Dr Dionysios Veronikis withdrew his offer. The clinician said he did not have time for games. Ms Sturgeon said she was "not aware of evidence" that backed up the claims. World-leading mesh surgeon reveals months of talks have failed to fix his trip to Scotland despite minister's pledge. The world-leading surgeon who operated on Claire Daisley has voiced frustration at delays in his proposed trip to Scotland to train surgeons and help other mesh-damaged women.
Dionysios Veronikis has been in discussions with Scots medical officers since June when Health Secretary Jeane Freeman finally accepted his offer to help, six months after mesh victims urged her to invite him to Scotland. However, after months of fruitless discussion about arrangements, he has now written to Scottish Government’s deputy chief medical officer Terry O’Kelly, copying in Ms Freeman, to explain the lack of progress is nothing to do with him. A first step... Sufferers applaud the Health Secretary after she paves way to bring US mesh surgeon to Scotland. Health Secretary Jeane Freeman has paved the way for a top US surgeon to come to Scotland to carry out pioneering mesh removal operations.
The decision came after an emotionally charged meeting between mesh implant victims and Ms Freeman at the Scottish Parliament last Tuesday. The move means that US surgeon Dr Dionysios Veronikis, who has developed his own highly successful mesh implant removal procedures, should now be able to come here to carry out operations and train medical staff. Grieving family demand answers over great-grandmother’s mesh death. A grieving son whose mother was the first person in Scotland to have mesh implant surgery listed as an underlying cause of death is demanding answers from the regulator who approved the product.
Great-grandmother Eileen Baxter died in August from multiple organ failure after being admitted to hospital with internal bleeding, sickness and diarrhoea a week earlier. The 75-year-old previously underwent sacrocolopexy mesh repair – an implant to fix a pelvic organ prolapse. Her son Mark Baxter, from Loanhead in Midlothian, and his sister Audrey have met health secretary Jeane Freeman to discuss their mother’s death and the implications of the wider use of mesh. This led to the Scottish Government announcing a halt in the use of transvaginal mesh, with only a limited number of operations to be considered for women already on the waiting list. An investigation into Eileen Baxter’s death is being carried out by associate medical and nurse directors at NHS Lothian.
Government halts vaginal mesh surgery in NHS hospitals. The government has accepted the use of vaginal mesh implants to treat complications after childbirth should be stopped immediately to prevent further risk of “life-changing and life-threatening injuries” to women.
It would effectively suspend the use of vaginal mesh implants in NHS hospitals, which would represent a major victory for campaigners. It follows an independent inquiry, ordered in February by the health secretary, Jeremy Hunt, which concluded the surgery must be stopped until steps have been taken to mitigate the risks to patients. Julia Cumberlege, who chaired the review, said: “I have been appalled at the seriousness and scale of the tragic stories we have heard from women and their families. We have heard from many women who are suffering terribly. Their bravery and dignity in speaking out is deeply moving, and their sadness, anger, pain and frustration at what has happened to them and others has been compelling.
Arrêt immédiat de l'utilisation des implants maillés dans NHS Scotland – Nouvelles Du Monde. Arrêt immédiat de l'utilisation des implants maillés dans NHS ScotlandBelfastTelegraph.co.ukL’utilisation d’implants à mailles a été immédiatement arrêtée dans le NHS Scotland pour certaines conditions, a annoncé le secrétaire à la santé.
L’utilisation d’implants à mailles a été immédiatement arrêtée dans le NHS Scotland pour certaines conditions, a annoncé le secrétaire à la santé. Dans un communiqué à Holyrood, Jeane Freeman a déclaré qu'une interdiction effective serait en place jusqu'à l'introduction d'un nouveau «protocole d'utilisation restreinte». Cela garantirait que la procédure ne serait pas utilisée, sauf dans les circonstances les plus exceptionnelles, où il n’existait pas d’alternative clinique et c’était le souhait exprès de la femme concernée de recevoir des informations complètes.
Mme Freeman a mis à jour les MSP la semaine après qu’elle ait révélé que la mort d’Eileen Baxter, âgée de 75 ans, en août était liée à un implant à mailles. I wanted to have my vaginal mesh implant removed – but it left me PARALYSED. LOOKING into her newborn son Niall’s eyes, Gemma Mulcahy was smitten.
The former model and mum-of-two - who showcased clothes on This Morning as a teen - briefly forgot the “really tough” pregnancy and “horrific birth” she’d just endured. Gemma Mulcahy / Hook News “My hips were in agony and by 37 weeks it was so bad I could barely walk and had to be induced,” Gemma, also mum to Mhairi, six, recalled. Grieving son who lost mother to mesh set to meet health secretary.
The son of the first woman in Scotland to have mesh listed as an underlying cause of death is to meet with health secretary Jeane Freeman tomorrow to discuss his mother’s case.
Mark Baxter will go to Holyrood with his sister Audrey and mesh campaigner Neil Findlay MSP to discuss the circumstances surrounding the death of their mother Eileen who passed away on 27 August. Mrs Baxter, a great-grandmother, who was married to 79-year-old Chic, had multiple organ failure listed as directly leading to her death after being admitted to the Royal Infirmary of Edinburgh, with internal bleeding, sickness and diarrhoea. Mesh campaigners welcome Government ban but say the fight goes on. THE women who first gave a voice to Scotland’s mesh victims yesterday welcomed a ban announced by Holyrood ministers last week.
But the two courageous campaigners said their fight to halt the use of all plastic surgical mesh – which continues to be used on both men and women – will continue. Health Secretary Jeane Freeman last week said NHS boards have been told to stop using vaginal mesh in cases of pelvic organ prolapse and stress urinary incontinence but other types of mesh procedures will continue. Elaine Holmes, who launched the Hear Our Voice campaign six years ago, said: “Although this is a phenomenal victory for patient power, it’s sad it took so long for the Government to listen. “It concerns us mesh procedures are still being carried out, and we will continue our campaign until mesh is no longer used.” Immediate halt on use of mesh implants in NHS Scotland.
The use of mesh implants has been immediately halted in NHS Scotland for certain conditions, the health secretary has announced.
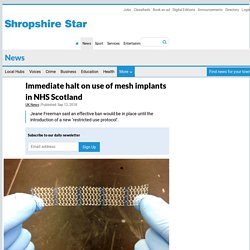
In a statement at Holyrood, Jeane Freeman said an effective ban would be in place until the introduction of a new “restricted use protocol”. This would ensure the procedure would not be used except in the most exceptional circumstances, where there was no clinical alternative and it was the express wish of the woman concerned after receiving full information.
Ms Freeman updated MSPs the week after it emerged the death of 75-year-old Eileen Baxter in August was linked to a mesh implant. “The instruction to halt is I believe a proportionate measure whilst a rigorous high vigilance restricted use protocol for any future practice is developed and put in place. “The lifting of this halt in use can only be considered once there is confidence that there’s sufficient evidence that the protocol will only be triggered in only the most limited of circumstances.” Ministerial Statement: Transvaginal Mesh : September 12, 2018. Health boards ordered to stop using mesh implants. Health boards in Scotland have been told to immediately stop using vaginal mesh implants. The move comes after campaigners called for the procedure to be halted after implants were implicated in the death of one woman and serious medical complications in many others.
Health minister Jeanne Freeman told MSPs on Wednesday that she had now taken the step of halting implant procedures across Scotland. In a statement at Holyrood, Ms Freeman said an effective ban would be in place until the introduction of a new "restricted use protocol". This would ensure the procedure would not be used except in the most exceptional circumstances, where there was no clinical alternative and it was the express wish of the woman concerned after receiving full information. Health Secretary to update parliament on mesh controversy. 'There is a horrible pattern of women not being listened to': Mhairi Black backs dying gran's fight for victims of mesh. MHAIRI BLACK is backing a dying mesh victim fighting to ensure the voices of other victims are heard.
The SNP MP hugged Marion McMillan and promised to do all she could to ensure her death will not be in vain. Marion, 70, who has been told she is “too ill” for medical help after doctors sent her to see a psychiatrist instead of for the scan that could have saved her life, is begging the NHS to monitor the health of mesh victims. The Paisley grandmother said: “Doctors kept denying the pain I was suffering in my back was because of mesh. They told me it was all in my head when in fact I was suffering from the early stages of cancer. “If they had sent me for a scan instead of sending me to a psychiatrist, I would have had a fighting chance. Ms Black, the MP for Paisley and Renfrewshire South, said: “It’s clear there is a horrible pattern emerging here of a large group of women reporting serious issues and nobody taking them seriously.”
First Do No Harm: Complications and Complaints will Change Pelvic Floor Surgery Using Mesh Implants in 2015. IntroductionIn a plenary session of the Scottish parliament on the 26th of June 2014 Alex Neil the Scottish cabinet minister for social justice gave a ministerial statement on the use of polypropylene (plastic) mesh devices in pelvic floor surgery. He described hearing of the “horrendous” complications suffered by some women after vaginal mesh surgery for incontinence and prolapse.
He related that traditional surgical methods without mesh had high failure rates of 25% or more and that the medicines and health care products regulatory agency (MHRA) had previously reported complication rates for mesh procedures of only 1-2.6%. He was now concerned as to the real incidence of serious complications and that many might be going unreported. Mesh implant listed as ‘cause of death’ for first time. A woman who underwent controversial mesh implant surgery is believed to be the first in Scotland to have the procedure listed as an underlying cause of death.
Eileen Baxter, 75, died on 27 August after being admitted to hospital the previous week with internal bleeding, sickness and diarrhoea. Multiple organ failure is listed as directly leading to her death, with sacrocolopexy mesh repair – an implant to fix a pelvic organ prolapse – listed as an underlying cause. Last night, Mrs Baxter’s son Mark, 52, said he was given the number for the legal department of the Royal Infirmary of Edinburgh by medical staff after he asked for a copy of his mother’s death certificate, which The Scotsman has seen.
Criminal probe call over vaginal mesh ‘dangers’ A CRIMINAL investigation must be held over the mesh implants scandal, an MSP says. Neil Findlay, Labour’s ex-shadow health secretary, has written to the head of Police Scotland, the country’s chief prosecutor and Justice Secretary Humza Yousaf urging investigators to open an inquiry into companies responsible for the devices. More than 20,000 women have been fitted with the implants since the 1990s, with surgeons using them to treat conditions including pelvic organ prolapse and incontinence. However, the procedure has led to debilitating pain for some, with those affected experiencing a range of problems like infections, bleeding and impaired mobility. Holyrood committee calls for a ban on mesh implants.
Fight for justice: "I want to die knowing other mesh victims might now be saved" Memos reveal mesh firms were warned 21 years ago that material should not be used on humans. Welsh Conservatives call for immediate suspension of surgical mesh procedures in Wales. The Independent Medicines and Medical Devices Safety Review has concluded that there must be an immediate pause in the use of surgical mesh for the treatment of stress urinary incontinence (SUI). Globetrotting Jill is bringing hope to mesh survivors. A globetrotting giraffe is bringing comfort to mesh-injured women the world over – and a Paisley mum-of-two is hoping it can work its magic on her.
Olive McIlroy had a mesh tape operation 10 years ago to “fix a leaky bladder,” and after suffering serious mesh complications, disability and chronic pain, she was forced to give up her work as a carer with Renfrewshire Council. But 60-year-old Olive, who now lives in Renfrew, had a beaming smile on her face when she met Jill the Giraffe for the first time. The woolly toy has clocked up more air miles than many frequent fliers and has visited mesh sufferers as far afield as the United States, Canada and New Zealand.
Now, Olive is the latest to offer ‘full board’ to Jill, and she was delighted to take temporary ownership of the cuddly companion. She told the Paisley Daily Express: “When I first heard about Jill the Giraffe I thought it was a great idea, because she has achieved so much in such a short space of time. Courageous Michele, who used her final days to warn fellow mesh victims of the risks, dies after cancer battle. How Wales plans to help nearly 7,000 women who have had a vaginal mesh. Vaginal mesh or tape should only be used “as a last resort”, a report has recommended, after it was discovered more than 400 patients in Wales have had it removed over the past decade.
The Welsh Government, which set up a group to review the use of mesh , says patients should only be given the treatment when all other options have been exhausted. 180504reporten. Brave mum dying from cancer speaks out after controversial mesh treatment left her too weak for chemo. Suspension of mesh surgery in Scotland means women need improved access to physiotherapy. In America, mesh victim wins £42m payout. In Scotland, ministers spend just £4500 on inquiry into the scandal - Sunday Post.
Mesh implant surgery 'decimated my quality of life' Mesh expert voices concerns over patient safety. The expert at the centre of a review group looking at the safety of mesh implants in Scotland has spoken for the first time about why he resigned. Dr Wael Agur told BBC Scotland he believed patient safety was being put at risk. The mesh implant scandal is a global catastrophe: Former health secretary Alex Neil calls for international summit to tackle escalating crisis - Sunday Post. Insight: Down but not out after mesh implant ‘whitewash’
‘We call ourselves Cagney and Lacey,” says Olive McIlroy of her friendship with Elaine Holmes. And there is something of the no-nonsense US cop duo about the women who have led the campaign against polypropylene transvaginal mesh implants in Scotland. The blend of vulnerability and bravado, maybe; and the refusal to be crushed by adversity. Along with hundreds of other women in Scotland – and tens of thousands across the world – Holmes and McIlroy suffered devastating complications after the mesh implants were used to treat Stress Urinary Incontinence (SUI), a problem common amongst middle-aged women. Pelvic mesh: Suffer in silence. Top surgeon says doctors are complicit in mesh scandal and it stains reputation of whole medical profession. A LEADING surgeon has warned doctors’ reputations will be ruined over the mesh scandal. Mark Slack, one of the most eminent consultants in his field, said “history will judge harshly” surgeons involved in the treatment which ruined the lives of hundreds of thousands of women.
Slack, head of gynaecology at Addenbrooke’s Hospital in Cambridge, condemned mesh manufacturers for what he has called a “tragedy created by greed”. And he claimed the fallout from the scandal, which has wrecked the health of more than 400 women in Scotland alone, will “stain” doctors who championed treatments which the Scottish Government have now virtually banned after a Sunday Mail campaign. Your horror stories of post baby op that can ruin lives. Probe to examine possible conflicts of interest in troubled mesh implant inquiry - Sunday Post.
Minister says expert will look at mesh implants review. Mesh survivors shall see the justice they deserve. Alex Neil 'disturbed and disappointed ' by the outcome of the mesh implants inquiry he established. Transvaginal Mesh Implants - Scottish Parliament: 30th March 2017. Mesh implant campaigners 'betrayed' by report publication. Mesh scandal report findings have ‘no credibility’ according to former Health Secretary Alex Neil - Sunday Post. Medical expert quits mesh implant review group. ‘Betrayed’ victims quit mesh implants inquiry - Sunday Post. Scottish MESH Survivors, News and Media Scotland, England, Worldwide. Mesh implants still used in Scotland despite suspension call. Mesh implants campaigners demand probe after US lawyer says medical firm used fake materials. PressReader.com - Connecting People Through News. Pelvic Mesh Implant Procedures Halted in Scotland - Mesh Medical Device Newsdesk.
Public Petitions Committee - Scottish Parliament: 8th March 2016. Scottish MESH Survivors. Mesh implants review recommends better training for medical staff (From Herald Scotland) Three of the four mesh procedures routinely used in Scottish hospitals will no longer be recommended. Physios should have a greater role in supporting mesh implant patients, enquiry finds. Celebrity Comments - Scottish MESH Survivors. Motions, Questions and Answers Search. 00486661. Government facing massive payout if women aren't warned about mesh implants.
Scottish MESH Survivors. Plastic mesh implants could be causing horrific health problems for Scots women. Scottish MESH Survivors, News and Media Scotland, England, Worldwide. Mesh surgery scandal: Health watchdog accused of backing procedures without fully understanding risks. Health Secretary says mesh surgery only to happen with 'fully informed consent' of patient. Support for mesh implant survivors - Government World Magazine. PE01517: Polypropylene Mesh Medical Devices - Getting Involved : Scottish Parliament. Motions, Questions and Answers Search. PE01517: Polypropylene Mesh Medical Devices - Getting Involved : Scottish Parliament. Patients in disbelief as mesh company sales reps were allowed in theatre while surgeons carried out intimate operations. Live: Scottish Parliament 24 February 2015 - BBC News. Mesh implants campaigners give evidence to MSPs.
Women urge MSPs to act over mesh implants. Ntroversial mesh implants to be banned by Dumfries and Galloway health board. Mesh surgery scandal: Health minister Alex Neil calls for urgent review of controversial treatment. PE01517: Polypropylene Mesh Medical Devices - Getting Involved : Scottish Parliament. Scottish Health Secretary Alex Neil requests mesh implant suspension. Mesh surgery scandal: Operation victims launch advice and support website. Mesh Surgery scandal: Exercise therapies costing less than £130 could have saved victims from agony of op.