

Chem1 Online Textbook Main Menu. Acid-base chemistry can be extremely confusing, particularly when dealing with weak acids and bases.

This set of lessons presents an updated view of the Brønsted-Lowry theory that makes it easy to understand answers to common questions: What's the fundamental difference between a strong acid and a weak acid? Can acid A neutralize base B? Why are some salts acidic and others alkaline? How do buffers work? What governs the shapes of titration curves? This complete rewrite of the previous 1996 pdf document is now organized into seven lessons covering all aspects of the subject. Chemistry 101 - Introduction and Index of Topics. By Anne Marie Helmenstine, Ph.D.
Updated March 16, 2016. Welcome to the world of Chemistry 101! Chemistry is the study of matter. Like physicists, chemists study the fundamental properties of matter and they also explore the interactions between matter and energy. Chemistry is a science, but it is also used in human communication and interaction, cooking, medicine, engineering, and a host of other disciplines.
11th Grade High School Chemistry Topics. Chemistry First. Table of Contents Printable Periodic Table Web Periodic Table Element List Conversion Factors.

Basics: Scientific Notation. Actions and Reactions. Upload ed.ted.com.
Reactions. High School Laboratory Safety Certification Course. The Flinn Scientific High School Laboratory Safety Course is a comprehensive seven-hour online safety training program.
The 45 separate video chapters that make up the course may be viewed either individually or as part of a certification sequence. In order to become “Flinn Certified” you must watch all 45 videos in sequence and complete a series of the unit assessments to document the safety training. If you choose to view the videos individually you will have the freedom to skip around and select any video of interest.
You will always have the ability to switch between viewing individual video chapters and participating in the certification course in the “My Progress” portion of your “My Account” page. Chapter notes will be available to print on that specific video player page. Home Chemistry: Experiments. We recently tried a new and improved version of the lava lamp project from several years ago.
It worked great! Here's how we did it: Tools and Materials: • Clean recycled soda or water bottle, label removed, 16 oz or larger • Water • Food coloring • Baby oil, at least one 12 oz bottle • Effervescent antacid tablets (such as Alka-Seltzer) • LED push light (or make a light-up base from a recycled jar or can, some extra bright LED bulbs, coin batteries, and tape) • Plastic plate or other protection for table • Cooking (meat) thermometer (optional) Step 1: Prepare the lava lamp bottle. The first step in building your homemade lava lamp is to find a tall, thin, clear bottle.
Step 2: Prepare the Bottom Layer of Colored Water. Chem Academy. Quarks and Quirks. DONE!
( Almost.) Aside from some of the labs near the end of the course and a “living books” assignment yet to be wiggled into the schedule, this syllabus is complete. Teacher Resources. Teacher Programs JLab Science Activities for Teachers (JSAT) - An afternoon science program for 5th, 6th and 8th grade teachers.
[Program Dates: September 2016 - May 2017] Physical Science Investigation - Jefferson Lab (PSI: JLab) - A week long summer program for teachers! [Application Deadline: June 30th, 2016] Teacher Night at Jefferson Lab - Teacher Night will be held on April 13th, 2016. Education Events. Chemistry. Chemistry.org: Science that Matters: American Chemical Society. Strange Matter: Processing. Periodic Table of Elements: Los Alamos National Laboratory. The Comic Book Periodic Table of the Elements. Chemical element games - periodic table info.
Adventures in Chemistry - American Chemical Society. Chemistry for Kids - Fun Experiments, Free Games, Cool Projects, Science Online. Reactions. Battle of the Slime! ChemMatters Magazine. Chemicool. Here’s a look at the remarkable properties of ten of the periodic table’s elements.
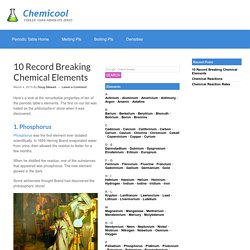
The first on our list was hailed as the philosophers’ stone when it was discovered. 1. Phosphorus Phosphorus was the first element ever isolated scientifically. In 1669 Hennig Brand evaporated water from urine, then allowed the residue to fester for a few months. When he distilled the residue, one of the substances that appeared was phosphorus. Some alchemists thought Brand had discovered the philosophers’ stone!
The Alchemist by Joseph Wright depicts Hennig Brand’s discovery of phosphorus. 2. Carbon forms more compounds than any other element. Carbon’s ability to bond with other elements makes it the ideal basis for life, such as this excited gull. 3. Gold is the most ductile, or drawable, of all metals. Here’s a 100 gram gold bar, shown with a pencil for scale. The Particle Adventure. General Chemistry Online. Common Compound Library A searchable database of over 800 common compound names, formulas, structures, and properties.

Companion Notes Hyperlinked notes and guides for first semester general chemistry. Construction Kits Flash-based kits for building chemical formulas, names, equations, and problem solutions. Articles Featured articles, books, and tutorials. Toolbox Interactive graphing, popup tables, and calculators. Tutorials Index of self-guided tutorials, quizzes, and drills on specific topics. Chemical Science Resources. The Periodic Table of Videos - University of Nottingham. Chemistry-Periodic Table and Atomic Theory on Pinterest. Rader's CHEM4KIDS.COM - Chemistry basics for everyone!