

Shiva Star. Shiva Star, originally just SHIVA, is a high-powered pulsed-power research device located at the Air Force Research Laboratory on the Kirtland Air Force Base in Albuquerque, New Mexico.
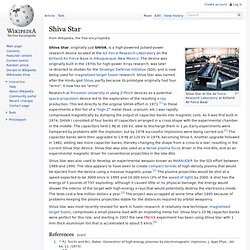
The device was originally built in the 1970s for high-power X-ray research, was later re-directed to studies for the Strategic Defense Initiative (SDI), and is now being used for magnetized target fusion research. Kinetic isotope effect. Kinetic isotope effect (KIE) refers to the change in the rate of a chemical reaction upon substitution of an atom in the reactants with one of its isotopes.
Formally, it is defined as the ratio of rate constants for the reactions involving the light (kL) and the heavy (kH) isotopically substituted reactants: For example, in the following nucleophilic substitution reaction of methyl bromide with cyanide, the kinetic isotope effect of the methyl carbon, in this case defined as k12/k13, was found to be 1.082 ± 0.008.[1][2] Muons take kinetic isotope effects to extremes.
Quantum mechanics. Wavefunctions of the electron in a hydrogen atom at different energy levels.
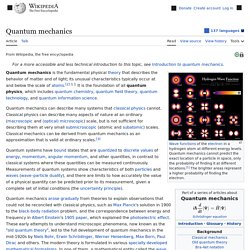
Quantum mechanics cannot predict the exact location of a particle in space, only the probability of finding it at different locations.[1] The brighter areas represent a higher probability of finding the electron. Quantum mechanics (QM; also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of atoms and subatomic particles.[2] Quantum mechanics gradually arose from theories to explain observations which could not be reconciled with classical physics, such as Max Planck's solution in 1900 to the black-body radiation problem, and from the correspondence between energy and frequency in Albert Einstein's 1905 paper which explained the photoelectric effect.
History[edit] In 1838, Michael Faraday discovered cathode rays. Where h is Planck's constant. Coulomb potential. Dirac equation. In particle physics, the Dirac equation is a relativistic wave equation derived by British physicist Paul Dirac in 1928.
In its free form, or including electromagnetic interactions, it describes all spin-½ massive particles, for which parity is a symmetry, such as electrons and quarks, and is consistent with both the principles of quantum mechanics and the theory of special relativity,[1] and was the first theory to account fully for special relativity in the context of quantum mechanics. Heisenberg Uncertainty Principle sets limits on Einstein's 'spooky action at a distance,' new research finds. Researchers have uncovered a fundamental link between the two defining properties of quantum physics.
[1004.2507] The uncertainty principle determines the non-locality of quantum mechanics. Some Frequently Asked Questions About Virtual Particles. [Physics FAQ] - [Copyright] Original by Matt McIrvin 1994.

Contents: What are virtual particles? How can they be responsible for attractive forces? Do they violate energy conservation? Do they go faster than light?
Fuels and thrusters. Electromagnetic Propulsion Ships, Submarines: patents & articles. Biefeld–Brown effect. There is renewed focus on the Biefield–Brown effect with the spread in interest in high-voltage powered experimental flying devices known as ionocraft or lifters.
[citation needed] Effect analysis[edit] The effect is generally believed to rely on corona discharge, which allows air molecules to become ionized near sharp points and edges. Usually, two electrodes are used with a high voltage between them, ranging from a few kilovolts and up to megavolt levels, where one electrode is small or sharp, and the other larger and smoother. The most effective distance between electrodes occurs at an electric potential gradient of about 10 kV/cm, which is just below the nominal breakdown voltage of air between two sharp points, at a current density level usually referred to as the saturated corona current condition.
Electrodynamic Space Thruster. Patent Pending PCT/IB2010/052975 1.

Presentation The Electrodynamic Space Thruster is a propulsion system designed by Moacir L. Ferreira Jr. in order to produce propulsive force in the outer space, using a sequenced pattern of phase-shifted electric oscillations, similarly to a linear AC motor, running much faster, creating sideway electrodynamic drag, consequently, producing an astonishing acceleration without infringing the classical laws of physics (action-reaction, action-at-a-distance). See also: Phase-shift Plasma Turbine 2. Propulsion in space occurs generally by expelling reaction mass, in accordance with Newton's third law of motion, commonly paraphrased as: "For every action force there is an equal, but opposite, reaction force" or in other words "action and reaction are always equal and opposite". 3.
Fischer–Tropsch process. The Fischer–Tropsch process is a collection of chemical reactions that converts a mixture of carbon monoxide and hydrogen into liquid hydrocarbons.

It was first developed by Franz Fischer and Hans Tropsch at the "Kaiser-Wilhelm-Institut für Kohlenforschung" in Mülheim an der Ruhr, Germany in 1925. The process, a key component of gas to liquids technology, produces a synthetic lubrication oil and synthetic fuel, typically from coal, natural gas, or biomass.[1] The Fischer–Tropsch process has received intermittent attention as a source of low-sulfur diesel fuel and to address the supply or cost of petroleum-derived hydrocarbons. [edit] The Fischer–Tropsch process involves a series of chemical reactions that produce a variety of hydrocarbons, ideally having the formula (CnH(2n+2)). The more useful reactions produce alkanes as follows: (2n + 1) H2 + n CO → CnH(2n+2) + n H2O where n is typically 10-20. Fischer Tropsch intermediates and elemental reactions Feedstocks: gasification[edit] Fischer-Tropsch Archive. How to turn seawater into jet fuel - tech - 18 August 2009.
Faced with global warming and potential oil shortages, the US navy is experimenting with making jet fuel from seawater.
Navy chemists have processed seawater into unsaturated short-chain hydrocarbons that with further refining could be made into kerosene-based jet fuel. But they will have to find a clean energy source to power the reactions if the end product is to be carbon neutral. The process involves extracting carbon dioxide dissolved in the water and combining it with hydrogen – obtained by splitting water molecules using electricity – to make a hydrocarbon fuel.
Fischer Tropsch Synthesis.
Ultracapacitors Make City Buses Cheaper, Greener. (PhysOrg.com) -- A fleet of 17 buses near Shanghai has been running on ultracapacitors for the past three years, and today that technology is coming to the Washington, DC, for a one-day demonstration. Chinese company Shanghai Aowei Technology Development Company, along with its US partner Sinautec Automobile Technologies, predict that this approach will provide an inexpensive and energy efficient way to power city buses in the near future. The biggest advantage of ultracapacitors is that they can fully recharge in less than a minute, unlike lithium-ion batteries which can take several hours.
The downside of ultracapacitors is that they currently have a very short range, providing a distance of only a few miles, due to the fact that ultracapacitors can store only about 5% of the energy that lithium-ion batteries can hold. Although their short range makes ultracapacitors impractical for cars, city buses have to stop frequently anyway. More information: Sinautec via: Technology Review.
Mitä maito on? FEDIOL.