

ChemCollective. L'Osmosi - Lachimica.video. Quimilab - Alumno. Laboratorio de química 1. Model Science Software Products - Model ChemLab. "Insegnare le procedure di un laboratorio chimico attraverso un laboratorio virtuale su di un personal computer è un concetto straordinario.
Gli studenti non devono sottostare a limiti di tempo e spazio e gli istituti scolastici che non dispongono dei mezzi fisici o economici per mantenere un verso laboratorio chimico accoglieranno con entusiasmo quest’alternativa. Model ChemLab è un semplice strumento. È compatibile con i sistemi operativi Windows e Macintosh, i più diffusi negli ambienti scolastici. L’utilizzo è molto intuitivo e facile da imparare. Chemistry Archives - The Lab. Dynamic Periodic Table. Educational Videos. Search and share chemistry. Chemistry resources for Teachers and Students. The Virtual Molecular Model Kit. Chemguide: helping you to understand Chemistry.
Practical Chemistry. This website is for teachers of chemistry in schools and colleges.
It is a collection of experiments that demonstrate a wide range of chemical concepts and processes. Some of the experiments can be used as starting-points for investigations or for enhancement activities. Many have links to carefully selected further reading and all include information and guidance for technicians. Chemistry is a practical science. The Photographic Periodic Table of the Elements. Www.chem.iastate.edu/group/Greenbowe/sections/projectfolder/flashfiles/reaction/bonding1.swf. Dynamic Table En+Spa. Science Experiments. Physics for Kids - Free Games, Fun Experiments, Activities, Science Online. Amusement Park Physics. Fysikaliska Leksaker. Light and Shadows Interactive Science Revision - Games and Activities. Light. Cyberphysics, the revision website. Synvillor, syn, optik, öga, optiska illutioner. History of the Periodic Table.
Figure 4-2.
Dmitri Mendeleev Created the Periodic Table That We Still Use Today © The Regents of the University of California, Lawrence Berkeley National Laboratory, 2010. View More Figure 4-2. Dmitri Mendeleev Created the Periodic Table That We Still Use Today Born in Siberia, Russia, Mendeleev was the youngest of 14 children. In the early 19th century, a scientist named Johann Döbereiner (1780–1849) noticed that strontium's atomic mass fell exactly between the atomic masses of calcium and barium. The most remarkable aspect of Mendeleev's periodic table, and the reason why his name has endured, is that Mendeleev left spaces open for elements that were not yet discovered.
Figure 4-3. . © Public Domain. View More Figure 4-3. Dmitri Mendeleev organized the elements by atomic weight for his table. Because the proton, the neutron, and the electron had not yet been discovered, Mendeleev initially sorted the elements by atomic mass. Periodic table activity. The Compound Interest Periodic Table of Data. Today’s graphic is one that I’ve been working on over the past couple of weeks.

Every chemistry classroom has a Periodic Table, but it’s often a drab affair; considering it’s one of the cornerstones of chemistry, I thought I’d attempt to produce a more dynamic looking version. The result is the above table – each element is shown within a circle that contains a variety of data pertaining to that element. The data shown for each element is as follows: Melting point in degrees celsius.Boiling point in degrees celsius.First ionisation energy in kilojoules per mole.Atomic radius in angstroms.Electronegativity (on the Pauling scale).Density (in grams per centimetre cubed (g/L for gases)). This data provided is provided mainly with A Level students in mind, hence the use of ˚C for temperature over Kelvin, and kilojoules per mole for ionisation energy over electron volts. The first version also includes a diagram to show the order in which electron orbitals fill. Using the Periodic Table to Determine Oxidation States by SAS® Curriculum Pathways®
The genius of Mendeleev’s periodic table - Lou Serico. EJERCICIOS Y EJE NOMENCLATURA. Questions and Answers. What is chemical energy? Chemical energy Chemical Energy is energy stored in the bonds of chemical compounds (atoms and molecules).

It is released in a chemical reaction, often producing heat as a by product (exothermic reaction). Batteries, biomass, petroleum, natural gas, and coal are examples of stored chemical energy. Usually, once chemical energy is released from a substance, that substance is transformed into an entirely new substance. Kinetic and Potential Energy. Kinetic energy is energy possessed by a body by virtue of its movement.
Potential energy is the energy possessed by a body by virtue of its position or state. While kinetic energy of an object is relative to the state of other objects in its environment, potential energy is completely independent of its environment. Hence the acceleration of an object is not evident in the movement of one object, where other objects in the same environment are also in motion. For example, a bullet whizzing past a person who is standing possesses kinetic energy, but the bullet has no kinetic energy with respect to a train moving alongside. Welcome to NanoSpace! Lezioni di chimica generale ed inorganica - Il legame chimico. Chemistry. The Periodic Table of Videos - University of Nottingham.
Chimica-online: risorse didattiche per lo studio online della Chimica. Chemistry Labs - OnlineLabs.in - Virtual laboratory simulations for science education. Below is a list of freely available online chemistry lab resources, including general chemistry and organic chemistry simulations.
Virtual Lab Simulator – From The ChemCollective. It started as the IrYdium Project’s Virtual Lab. There is extensive documentation for the lab, including an instruction video. The Java lab applet is translated into Spanish, Portuguese and CatalanVirtlab: A Virtual Laboratory – Registration required, but you can join Virtlab for freeVirtual Chemistry Experiments – Chemistry and Physics applets and phylsets from Davidson University.
Some exercises are currently broken and others (like the Chemical Equilibria exercises) work fineDartmouth ChemLab – This site has some very good interactive virtual labs plus a terrific interactive periodic tableVirtual Chemistry Lab – From the University of Oxford. The following chemistry laboratory simulations and educational learning exercises are available for a fee. Commercial products Return to the List of Subjects. Download Free Science Activities, Access Chemistry Multimedia, Find Information on Workshops. Free chemistry Animations- Learnerstv.com. Jefferson Lab.
Chemistry IYC 2011 and Beyond: Education. Chemistry Now. Chemistry resources for Teachers and Students. Practical Chemistry. Chemistry Activities and Games Free Downloads. The Molecular View of Equilibrium. Text below taken from Kinetic Theory of Gases: Molecular Speeds Other sections state that increasing the temperature increases the speeds at which molecules move.
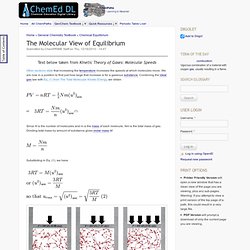
We are now in a position to find just how large that increase is for a gaseous substance. Combining the ideal gas law with Eq. (1) from The Total Molecular Kinetic Energy, we obtain. Distillation. Laboratory display of distillation: 1: A source of heat 2: Still pot 3: Still head 4: Thermometer/Boiling point temperature 5: Condenser 6: Cooling water in 7: Cooling water out 8: Distillate/receiving flask 9: Vacuum/gas inlet 10: Still receiver 11: Heat control 12: Stirrer speed control 13: Stirrer/heat plate 14: Heating (Oil/sand) bath 15: Stirring means e.g.
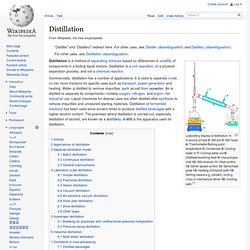
(shown), boiling chips or mechanical stirrer 16: Cooling bath.[1] History[edit] Clear evidence of the distillation of alcohol comes from the School of Salerno in the 12th century.[2][5] Fractional distillation was developed by Tadeo Alderotti in the 13th century.[6] A retort. GCSE Bitesize - Structure and bonding. Build an Atom. How atoms bond - George Zaidan and Charles Morton. GCSE Bitesize - Acid, bases and salts. RSC Aluminium Extraction - video. Electroplating animation.wmv. AQA Unit 2 Chemistry. Acids, bases and salts Lesson 4: Acid + Alkali. AQA Unit 2 Structure and Bonding Lesson 9 Properties of simple covalent molecules.
The Physics Classroom. Periodic Videos. S experimentfabrik. FISICA DINAMICA: Algodoo. Algodoo is now available as a free download.
If you want to support the development of Algodoo you can donate via PayPal below. You can also purchase it from the App Store with all its benefits. Or, you can simply download it for free below, either way is fine with us. Download for Mac Support the development of Algodoo for the Mac and buy your copy from the Mac App Store. Download for iPad Support the development of Algodoo for the iPad and buy your copy from the App Store. Build an Atom. Newton's 3 Laws of Motion. Electron Distributions. Instructions Before viewing an episode, download and print the note-taking guides, worksheets, and lab data sheets for that episode, keeping the printed sheets in order by page number. During the lesson, watch and listen for instructions to take notes, pause the video, complete an assignment, and record lab data. See your classroom teacher for specific instructions.
Note Taking Guide. Electronic configuration of atoms using Aufbau, Pauli's principle and Hund's rule - Chemistry. History of the Periodic Table. Metales, no metales y metaloides. Periodic Table of the Elements by WebElements. Visual Elements Periodic Table. Periodic Table – Royal Society of Chemistry. Dark matter is the thread connecting galaxy clusters. Simulations of the Universe on the largest scales show an unexpected resemblance to nerve cells in the human brain, with galaxy clusters playing the role of the cell body and thinner filaments of matter linking them like axons. Galaxy surveys (such as the Sloan Digital Sky Survey, or SDSS) show that galaxies do cluster like our simulations predict.
But the filaments that should connect them have been harder to find. Most of the mass in the Universe is dark matter—material that neither emits nor absorbs light—and filaments are predicted to be mostly dark matter: no galaxies, little hot gas. Einstein's general theory of relativity, however, tells us mass affects the path of light, and a group of astronomers have identified a dark matter filament by measuring this effect. Jörg P. These observations lend strong support to the theory that the Universe is built on a web of dark matter that has drawn in visible structures like galaxies and clusters.
Cf10371.pdf. Balancing Chemical Equations. 27 Reacciones Químicas Más Impresionantes. 10 Incredible Chemical Reaction GIFs Explained. We encounter thousands of chemical reactions every day: plants use them in photosynthesis, metals rust over time, and combustion reactions provide us with heat and light, among thousands of other daily uses. Chemical reactions occur when reactants transform into new substances, called products, through creating and breaking bonds between atoms. Sometimes the process creates some pretty wild effects. Check out our top 10 chemical reactions below: 1) Disintegration (Mercury Reacts with Aluminum) Image credit: Theodore Gray via Youtube When aluminum rusts, it creates a protective oxide layer that prevents the aluminum atoms underneath from further rusting.
Biotecnología. Balancing Chemical Equations - Phet. Build an Atom - Phet. Topics Atoms Atomic Structure Isotope Symbols Atomic Nuclei Description Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Balancing Act! - Balancing equations activity. Balancing Chemical Equations. How atoms bond - George Zaidan and Charles Morton. Chemistry Formula Practice Lite. Build a Molecule - Phet. How atoms bond - George Zaidan and Charles Morton. Enlace covalente - animación ETSII. Enlace Ionico - animación ETSII. Tipos de enlaces quimicos yeb01. Youtube. ¿QUE SON LOS ENLACES QUÍMICOS? ¿QUÉ TIPOS DE ENLACES QUÍMICOS EXISTEN? ENLACES QUÍMICOS EA)