

Matter and Motion. Matter: Solutions. Before we dive into solutions, let's separate solutions from other types of mixtures.

Beer's Law Lab - Solute, Solvent, Solutions. Why Do Oil and Water Not Mix. Matter: Changing States. All matter can move from one state to another.

It may require extreme temperatures or extreme pressures, but it can be done. Sometimes a substance doesn't want to change states. You have to use all of your tricks when that happens. To create a solid, you might have to decrease the temperature by a huge amount and then add pressure. For example, oxygen (O2) will solidify at -361.8 degrees Fahrenheit (-218.8 degrees Celsius) at standard pressure. Matter: States of Matter. We look at five states of matter on the site.

Solids, liquids, gases, plasmas, and Bose-Einstein condensates (BEC) are different states of matter that have different physical properties. Solids are often hard, liquids fill containers, and gases surround us in the air. Each of these states is also known as a phase. How does matter change from one state to another? Elements and compounds can move from one state to another when specific physical conditions change.
Why is this funny? - Curious Train. Particle Model of Matter. The Difference Between Atoms, Molecules, And Particles. “We must be clear that when it comes to atoms, language can be used only as in poetry.
The poet, too, is not nearly so concerned with describing facts as with creating images and establishing mental connections.” —Niels Bohr In A Nutshell Atoms are the smallest pieces of matter; they are made of particles (protons and electrons). When atoms are grouped together, these groups are called molecules (the smallest pieces of compounds). Thermal expansion. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature,[1] through heat transfer.
When a substance is heated, its particles begin moving more and thus usually maintain a greater average separation. Materials which contract with increasing temperature are unusual; this effect is limited in size, and only occurs within limited temperature ranges (see examples below). The degree of expansion divided by the change in temperature is called the material's coefficient of thermal expansion and generally varies with temperature.
Overview[edit] Predicting expansion[edit] If an equation of state is available, it can be used to predict the values of the thermal expansion at all the required temperatures and pressures, along with many other state functions. Contraction effects[edit] Thermal Expansion. Will melting ice cause water level to rise? Why solid expand on heating? - Answers.com. Diffusion and Osmosis. How Diffusion Works. Compression. Flash point. The flash point is not to be confused with the autoignition temperature, which does not require an ignition source, or the fire point, the temperature at which the vapor continues to burn after being ignited.
Neither the flash point nor the fire point is dependent on the temperature of the ignition source, which is much higher. What is the freezing point of candle wax? - Answers.com. Fire things. Solder. A soldered joint used to attach a wire to the pin of a component on the rear of a printed circuit board.
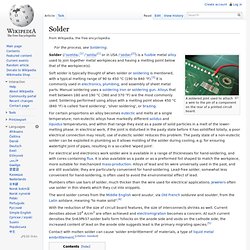
Solder (/ˈsoʊldə/,[1] /ˈsɒldə/[1] or in USA /ˈsɒdər/[2]) is a fusible metal alloy used to join together metal workpieces and having a melting point below that of the workpiece(s). Soft solder is typically thought of when solder or soldering is mentioned, with a typical melting range of 90 to 450 °C (190 to 840 °F).[3] It is commonly used in electronics, plumbing, and assembly of sheet metal parts. Manual soldering uses a soldering iron or soldering gun.
Alloys that melt between 180 and 190 °C (360 and 370 °F) are the most commonly used. What does fire produce. Phosphorus. The first form of elemental phosphorus to be produced (white phosphorus, in 1669) emits a faint glow upon exposure to oxygen – hence its name given from Greek mythology, Φωσφόρος meaning "light-bearer" (Latin Lucifer), referring to the "Morning Star", the planet Venus.
The term "phosphorescence", meaning glow after illumination, originally derives from this property of phosphorus, although this word has since been used for a different physical process that produces a glow. The glow of phosphorus itself originates from oxidation of the white (but not red) phosphorus— a process now termed chemiluminescence.
The vast majority of phosphorus compounds are consumed as fertilisers. Other applications include the role of organophosphorus compounds in detergents, pesticides and nerve agents, and matches.[5] Phosphorus is essential for life. Characteristics Physical P4 molecule White phosphorus and related molecular forms White phosphorus White phosphorus exposed to air glows in the darkness Isotopes. Flash point. Autoignition temperature. The autoignition temperature or kindling point of a substance is the lowest temperature at which it will spontaneously ignite in a normal atmosphere without an external source of ignition, such as a flame or spark.
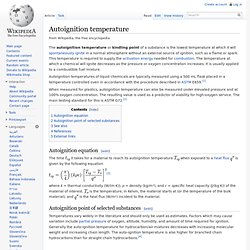
This temperature is required to supply the activation energy needed for combustion. The temperature at which a chemical will ignite decreases as the pressure or oxygen concentration increases. Paraffin Wax Flash Point. What are the ingredients in paraffin wax candles? - Answers.com. Ground-coupled heat exchanger. A ground-coupled heat exchanger is an underground heat exchanger that can capture heat from and/or dissipate heat to the ground.
They use the Earth's near constant subterranean temperature to warm or cool air or other fluids for residential, agricultural or industrial uses. If building air is blown through the heat exchanger for heat recovery ventilation, they are called earth tubes (also known as earth cooling tubes or earth warming tubes) in Europe or earth-air heat exchangers (EAHE or EAHX) in North America.
These systems are known by several other names, including: air-to-soil heat exchanger, earth channels, earth canals, earth-air tunnel systems, ground tube heat exchanger, hypocausts, subsoil heat exchangers, thermal labyrinths, underground air pipes, and others. States of Matter Rap.