

Heat pump and refrigeration cycle. Thermodynamic heat pump cycles or refrigeration cycles are the conceptual and mathematical models for heat pumps and refrigerators.
A heat pump is a machine or device that moves heat from one location (the 'source') at a lower temperature to another location (the 'sink' or 'heat sink') at a higher temperature using mechanical work or a high-temperature heat source.[1] Thus a heat pump may be thought of as a "heater" if the objective is to warm the heat sink (as when warming the inside of a home on a cold day), or a "refrigerator" if the objective is to cool the heat source (as in the normal operation of a freezer).
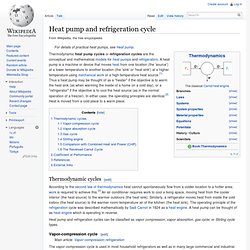
In either case, the operating principles are identical.[2] Heat is moved from a cold place to a warm place. Thermodynamic cycles[edit] Heat pump and refrigeration cycles can be classified as vapor compression, vapor absorption, gas cycle, or Stirling cycle types. Vapor-compression cycle[edit] Figure 1: Vapor-compression refrigeration Vapor absorption cycle[edit] Stirling engine. Alpha type Stirling engine.
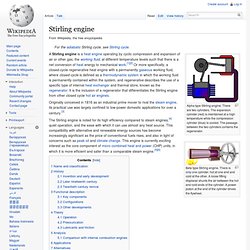
There are two cylinders. The expansion cylinder (red) is maintained at a high temperature while the compression cylinder (blue) is cooled. The passage between the two cylinders contains the regenerator. Beta type Stirling engine. There is only one cylinder, hot at one end and cold at the other. Originally conceived in 1816 as an industrial prime mover to rival the steam engine, its practical use was largely confined to low-power domestic applications for over a century.[3] The Stirling engine is noted for its high efficiency compared to steam engines,[4] quiet operation, and the ease with which it can use almost any heat source. Name and classification[edit] Robert Stirling was a Scottish minister who invented the first practical example of a closed cycle air engine in 1816, and it was suggested by Fleeming Jenkin as early as 1884 that all such engines should therefore generically be called Stirling engines.
History[edit] Invention and early development[edit] Ice. A glacier is made from ice, itself resulting from snow accumulation. Frozen water in the form of an ordinary (household) ice cube. The white zone in the center is due to tiny air bubbles. Ice is water frozen into a solid state. It can appear transparent or opaque bluish-white color, depending on the presence of impurities such as soil particles or air inclusions.
Ice is used for a wide range of applications including cooling, winter sports and the art of ice sculpting. The word is derived from Old English īs, which in turn stems from Proto-Germanic isaz. Characteristics Crystal structure of hexagonal ice. As a naturally-occurring crystalline inorganic solid with an ordered structure, ice is considered a mineral.[2] It possesses a regular crystalline structure based on the molecule of water, which consists of a single oxygen atom covalently bonded to two hydrogen atoms, or H-O-H. When ice melts, it absorbs as much energy as it would take to heat an equivalent mass of water by 80 °C.
Stirling cycle. This article is about the "adiabatic" Stirling cycle.
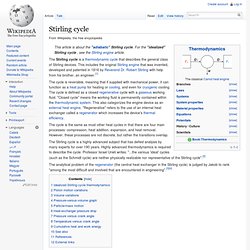
For the "idealized" Stirling cycle , see the Stirling engine article. The Stirling cycle is a thermodynamic cycle that describes the general class of Stirling devices. This includes the original Stirling engine that was invented, developed and patented in 1816 by Reverend Dr. Robert Stirling with help from his brother, an engineer.[1] The cycle is reversible, meaning that if supplied with mechanical power, it can function as a heat pump for heating or cooling, and even for cryogenic cooling. The cycle is the same as most other heat cycles in that there are four main processes: compression, heat addition, expansion, and heat removal. The Stirling cycle is a highly advanced subject that has defied analysis by many experts for over 190 years. Idealized Stirling cycle thermodynamics[edit] A pressure/volume graph of the idealized Stirling cycle.
Isothermal expansion. Piston motion variations[edit] A model of a four-phase Stirling cycle.