Online Books : "TIHKAL" - #18 4-HO-DMT
SYNTHESIS : To a solution of 0.50 g 4-acetoxyindole (see preparation in the recipe for 4-HO-DET) in 4 mL Et2O, that was stirred and cooled with an external ice bath, there was added, dropwise, a solution of 0.5 mL oxalyl chloride in 3 mL anhydrous Et2O. Stirring was continued for 0.5 h and the intermediate indoleglyoxylchloride separated as a yellow crystalline solid but it was not isolated. There was then added, dropwise, a 40% solution of dimethylamine in Et2O until the pH came to 8-9. The reaction was then quenched by the addition of 100 mL CHCl3, and the organic phase was washed with 30 mL of 5% NaHSO4 solution, with 30 mL of saturated NaHCO3, and finally with 30 mL of saturated brine. A suspension of 0.38 g LAH in 10 mL anhydrous THF was held in an inert atmosphere and vigorously stirred. Most of the early syntheses of psilocin and psilocybin employ the O-benzyl ether as a protecting group. DOSAGE : 10 - 20 mg, orally (as the indolol, the acetate or the phosphate) Another aside.
Btk. 176. § Kábítószer kereskedelem -
Kábítószer-kereskedelem 176. § (1) Aki kábítószert kínál, átad, forgalomba hoz, vagy azzal kereskedik, bűntett miatt két évtől nyolc évig terjedő szabadságvesztéssel büntetendő. (2) A büntetés öt évtől tíz évig terjedő szabadságvesztés, ha a bűncselekményt a) bűnszövetségben, b) hivatalos vagy közfeladatot ellátó személyként, e minőséget felhasználva vagy c) a Magyar Honvédség, a rendvédelmi szervek vagy a Nemzeti Adó- és Vámhivatal létesítményében követik el. (3) A büntetés öt évtől húsz évig terjedő vagy életfogytig tartó szabadságvesztés, ha a bűncselekményt jelentős mennyiségű kábítószerre követik el. (4) Az (1)-(3) bekezdés szerint büntetendő, aki az ott meghatározott bűncselekmény elkövetéséhez anyagi eszközöket szolgáltat. (5) Aki csekély mennyiségű kábítószert kínál vagy átad, a) az (1) bekezdésben meghatározott esetben vétség miatt két évig, b) a (2) bekezdés b)-c) pontjában meghatározott esetben egy évtől öt évig terjedő szabadságvesztéssel büntetendő. (6) Aki ca) kínál, átad, Kábítószeren:
Psilocybin
Psilocybin is Schedule I in the United States. This means it is illegal to manufacture, buy, possess, or distribute without a DEA license. Not available by prescription. Fresh and dried psilocybin mushrooms are considered containers of Psilocybin, making them illegal to possess as well. Addictive Potential: None Emergency Room Visits Yearly: Unknown Mandatory Minimum Sentence: Unknown Mechanism of Action: agonist of the serotonin receptors primarily at 5-HT2A and 5-HT2C Overview: Psilocybin is also known as 4-phosphoryloxy-N,N-dimethyltryptamine. Mushrooms containing psilocybin and psilocin are available fresh or dried and are typically taken orally. Pharmacology: Trip Reports: Side Effects and Adverse Reactions: Psilocybin and psilocin can produce muscle relaxation or weakness, ataxia, excessive pupil dilation, nausea, stomach pain, vomiting, and drowsiness. Articles: Research: E-books: Recommended In-Print Books: Other Informational Resources: Psilocybin: Investigator’s Brochure
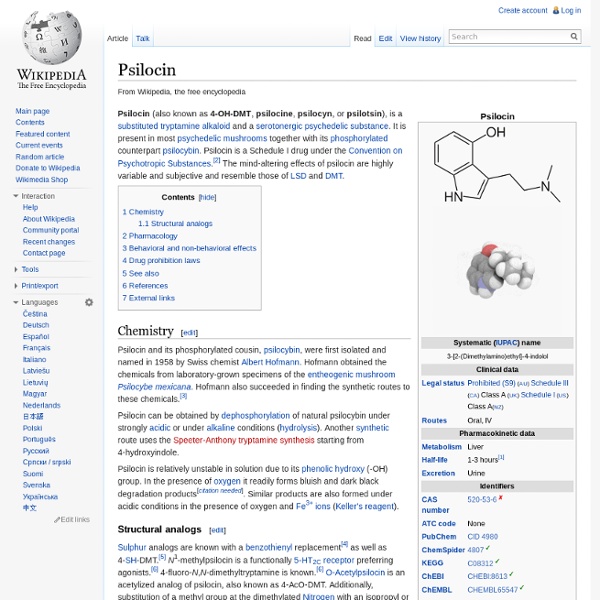
Psilocin
Psilocin is Schedule I in the United States. This means it is illegal to manufacture, buy, possess, or distribute without a DEA license. Not available by prescription. Addictive Potential: None Emergency Room Visits Yearly: Unknown Mandatory Minimum Sentence: Unknown Mechanism of Action: Agonist of the serotonin receptors, at 5-HT2A and 5-HT2C, among others (see a more detailed discussion below) Overview: Psilocin is also known as 4-hydroxy-N,N-dimethyltryptamine and 4-HO-DMT. Mushrooms containing psilocybin and/or psilocin are available fresh or dried and are typically taken orally. Pharmacology: The graph below (Ray, 2010) shows the affinity of psilocin for forty-two receptors, arranged in order of decreasing affinity (click the image to enlarge). As explained by Ray (2010), “The black vertical bar represents a 100-fold drop in affinity relative to the receptor with the highest affinity. Side Effects and Adverse Reactions: Research: Other Informational Resources:
Dr. Albert Hofmann | Psilocybin | Psilocin
Dr. Albert Hofmann, the brilliant Swiss chemist, philosopher, author, and retired Director of the Pharmaceutical-Chemical Research Laboratories of Sandoz Ltd., Basel, is best known for fathering his "problem child," LSD, on April 19, 1943. Thirteen years later, a brief article in a local paper caught his eye. The article stated that a researcher from America had traveled to southern Mexico and participated in a native ritual where mushrooms were consumed that produced strange visions. A year later Dr. Dr. In 1956, Moore, who was in reality a CIA operative specializing in the synthesis of psychoactive and chemical weapons for the CIA, offered R. Above: Molecular structures of LSD and psilocybin, written by Dr. Fortunately, Moore's efforts were unsuccessful and the honor, deservedly, went to Dr. Figure 1. Dr. Figure 2. Figure 3. Interestingly, in 1962 Dr. Now in his 90s, Dr. Above: Dr. Figure 4a. Figure 4b. Figure 5. Figure 6. Figure 7. Required Reading Aboul-Enein, H.
Mushroom Info
Our complete collection of psychedelic mushrooms information. Documents about experiencing shrooms safely and a great introduction to the Shroomery. An Introduction A document to welcome newcomers to the Shroomery and let them in on what's going on.
Psilocybin mushroom
Psilocybin mushrooms, also known as psychedelic mushrooms, are mushrooms that contain the psychedelic drugs psilocybin and psilocin. Common colloquial terms include magic mushrooms and shrooms.[1] Biological genera containing psilocybin mushrooms include Copelandia, Galerina, Gymnopilus, Inocybe, Mycena, Panaeolus, Pholiotina, Pluteus, and Psilocybe. About 40 species are found in the genus Psilocybe. Psilocybe cubensis is the most common psilocybin mushroom in subtropical areas and the black market. Psilocybin mushrooms have likely been used since prehistoric times and may have been depicted in rock art. History[edit] Early[edit] Archaeological evidence indicates the use of psilocybin-containing mushrooms in ancient times. Hallucinogenic species of the psilocybe genus have a history of use among the native peoples of Mesoamerica for religious communion, divination, and healing, from pre-Columbian times to the present day. Modern[edit] Occurrence[edit] Effects[edit] Sensory[edit]
Psilocybin
Psilocybin[nb 1] (/ˌsɪləˈsaɪbɪn/ SIL-ə-SY-bin) is a naturally occurring psychedelic compound produced by more than 200 species of mushrooms, collectively known as psilocybin mushrooms. The most potent are members of the genus Psilocybe, such as P. azurescens, P. semilanceata, and P. cyanescens, but psilocybin has also been isolated from about a dozen other genera. As a prodrug, psilocybin is quickly converted by the body to psilocin, which has mind-altering effects similar (in some aspects) to those of LSD, mescaline, and DMT. In general, the effects include euphoria, visual and mental hallucinations, changes in perception, a distorted sense of time, and spiritual experiences, and can include possible adverse reactions such as nausea and panic attacks. History[edit] Early[edit] Modern[edit] Albert Hofmann (shown here in 1993) purified psilocybin and psilocin from Psilocybe mexicana in the late 1950s. Occurrence[edit]