

Merck (Mosheim, France) € 25 million investment in Molsheim, France, will create 350 new jobsResponse to unprecedented global demand of key technologyProduction capabilities anticipated to be operational by end of 2021 Darmstadt, Germany, March 17, 2021 – Merck KGaA, Darmstadt, Germany, a leading science and technology company, today announced that it will add a single-use assembly production unit at its Life Science Center in Molsheim, France.
With the € 25 million investment, the company is accelerating its European expansion plans for this key technology, which is used for the production of Covid-19 vaccines and other lifesaving therapies. Molsheim will be the first site in Europe where the Life Science business sector manufactures the product. Further production sites are located in Danvers, Massachusetts, and Wuxi, China. The project was announced today in Molsheim in the presence of EU Commissioner Thierry Breton and the French Minister Delegate for Industry, Agnès Pannier-Runacher. Giant plastic bags (MilliporeSigma, Thermo Fisher) Merck Acquires AmpTec to Expand mRNA Capabilities for Vaccines, Treatments and Diagnostics.
TriLink. Sequence Optimization & Chemical Modifications While not technically part of the manufacturing process, sequence optimization and selection of chemical modifications are important to define at the start.
Strategic design of the mRNA sequence can enhance stability and translation in vivo, necessary qualities for an mRNA therapeutic. Both synonymous codons and modified NTPs can be utilized as strategies to deplete uridine content, thus reducing mRNA immunogenicity and improving translation. TriLink offers a wide catalog of modified NTPs for mRNA design, including pseudouridine triphosphate, N1-methylpseudouridine triphosphate, and 5-methoxyuridine triphosphate. Custom NTPs are also available. Plasmid Manufacturing Most mRNA therapeutics are produced by transcribing a linearized plasmid DNA template. mRNA Manufacturing In vitro transcription is a well-established method of synthesizing mRNA. Capping A 5’ cap structure is naturally added to mRNA synthesized in vivo.
Allele Biotech. Allele Biotechnology and SCM Lifescience Sign a Joint Research and Development Agreement to Develop iPSC-based Diabetes Treatment April 15th, 2019 -- San Diego-based Allele Biotechnology and Pharmaceuticals, Inc., officially announced a joint research and development agreement with South Korea’s SCM Lifescience Co., Ltd., for the development of diabetes therapies using pancreatic beta cells derived from induced pluripotent stem cells (iPSCs). iPSCs can be derived from adult human tissue cells yet share the developmental potentials of embryonic stem cells.

Currently Japan has 6 ongoing clinical trials utilizing this technology, but the rest of the world is now joining the movement with 4 clinical trials initiated outside of Japan in the past year. Since 2009, Allele Biotechnology has been developing clinically compatible technologies to produce high quality iPSCs and tissue-specific cells from iPSCs. Dr. Charles River Laboratories. At Charles River, we are committed to the conservation of this extraordinary animal that plays a role in everyday health and safety of people all over the world.
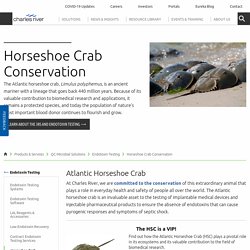
The Atlantic horseshoe crab is an invaluable asset to the testing of implantable medical devices and injectable pharmaceutical products to ensure the absence of endotoxins that can cause pyrogenic responses and symptoms of septic shock. The HSC is a VIP! Find out how the Atlantic Horseshoe Crab (HSC) plays a pivotal role in its ecosystems and its valuable contribution to the field of biomedical research. Read the Brochure In 1992, Dr. Mettler Toledo. General Electric.