

Future reading. Review - Vision in Drosophila: Seeing the World Through a Model's Eyes. Schematic anatomy. English [ edit ]
Drosophila Image Award - 2012. Subcellular Localization Profiling Reveals Synaptic Rab GTPases.
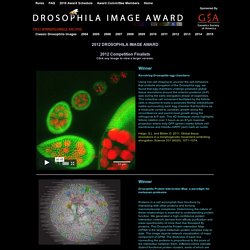
The compound eye and first optic ganglion of the fly: A light and electron microscopic study - PEDLER - 2011 - Journal of the Royal Microscopical Society. H1 neuron. The H1 neuron is located in the visual cortex of true flies of the order Diptera and mediates motor responses to visual stimuli.
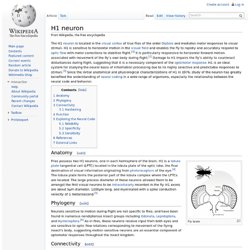
H1 is sensitive to horizontal motion in the visual field and enables the fly to rapidly and accurately respond to optic flow with motor corrections to stabilize flight. [ 1 ] It is particularly responsive to horizontal forward motion associated with movement of the fly’s own body during flight. [ 2 ] Damage to H1 impairs the fly’s ability to counteract disturbances during flight, suggesting that it is a necessary component of the optomotor response . H1 is an ideal system for studying the neural basis of information processing due to its highly selective and predictable responses to stimuli. [ 3 ] Since the initial anatomical and physiological characterizations of H1 in 1976, study of the neuron has greatly benefited the understanding of neural coding in a wide range of organisms, especially the relationship between the neural code and behavior.
HS coupling. Processing of Horizontal Optic Flow in Three Visual Interneurons of the Drosophila Brain. Flies rely heavily on visual motion information to navigate safely through the environment ( Borst and Haag 2002 ).

Once airborne, they use the characteristic flow fields caused by their self-motion to correct for deviations from a straight flight path. The precision and reliability of these so-called optomotor responses, combined with the small size of their brain, make flies an ideal organism to study the underlying neural circuitry ( Chan et al. 1998 ; Egelhaaf et al. 2003 ; Frye and Dickinson 2001 ; Götz 1964 ; Heisenberg et al. 1978 ).
Detailed anatomical maps describing the cell types of the optic lobes ( Fischbach and Dittrich 1989 ; Scott et al. 2002 ; Strausfeld 1976 ) are at hand. Disentangling the functional consequences of th... [Nat Neurosci. 2012. Interactive Reichardt Detectors. Move your mouse over the green receptors.

Instructions: Move your mouse over the green detectors above (you must have QuickTime 3.0 or better installed). You'll see that you get an "action potential" that stimulates the bell. 1 action potential isn't enough to sound the bell (below threshold), you need 2 arriving at about the same time to make the bell ring. To do that you must move your mouse at the right velocity. If it stops responding, try reloading this page. Krapp 1998 - Dendritic Structure and Receptive-Field Organization of Optic FlowProcessing Interneurons in the Fly. Segev and Borst - 2007 - Robust coding of flow-field parameters by axo-axonalgap junctions between fly visual interneurons. Supporting Information. Nature Neuroscience 2004 - neural mechnisms underlying complex receptieve field properties of motion-sesitive interneurons. Data bases. The Interactive Fly - A cyberspace guide to Drosophila development and metazoan evolution.
Virginia - Images of single labeled neurons using V3D to trace neurons. Genelia farm - 3d reconstruction - V3D. March 14, 2010 A Swiss Army Knife for Analyzing Three-Dimensional Images For centuries, anatomists have drawn illustrations of the body’s skeleton, nerve pathways, circulatory systems, and internal organs to help determine how the different parts work together.
Today, computers, microscopes and molecular genetics allow scientists to build more sophisticated, three-dimensional representations of living organisms. To create such models, researchers must synthesize information from thousands of detailed images that reveal different information: different magnifications of a structure or different biological characteristics, such as varying gene expression across cell types. Scientists at the Howard Hughes Medical Institute’s Janelia Farm Research Campus are unveiling a new software suite packed with useful tools for visualizing, analyzing, and measuring complex, three-dimensional biological and biomedical images.
Fly brain Atlas. Introduction (see below) Schematic Representations Major Brain Centres The Dissectable Brain Flybrain 3D Project Reduced Silver Sections.

NEURO LEX - Neuroscience lexicon : Vertical system neuron 1. Virtual Fly Brain: VFB Home. VS cell. FlyBase - Genes and Genomes. VS cells. ModelDB: Fly lobular plate VS cell (Borst and Haag 1996, et al. 1997, et al. 1999) 1999 - The Intrinsic Electrophysiological Characteristics of Fly Lobula Plate Tangential Cells: III. Visual Response Properties. 1997 - Encoding of Visual Motion Information and Reliability in Spikingand Graded Potential Neurons. 1996 - The intrinsic electrophysiological characteristics of fly lobula plate tangential cells: I. Passive membrane properties. Summary. Visual system. CH-cells. HS-cells. VS-cells. Quiz - Fly's visual syste. Passive membran parameters. IV-curve. NeuroMorph VS cells. Abstract.
Role of dendrites in sensory processing (Segev and Borst) Direction of motion could be computed by the dendritic tree. Branco - 2010, Science. ModelDB: Dendritic Discrimination of Temporal Input Sequences (Branco et al. 2010) E&I receptors located on dendritic branches HS and VS cells. Raghu SV[Author. Synaptic Organization of Lobula PlateTangential Cells in Drosophila.
Journal of Comparative Neurology Volume 502, Issue 4 , Article first published online: 29 MAR 2007 Options for accessing this content: If you have access to this content through a society membership, please first log in to your society website.

If you would like institutional access to this content, please recommend the title to your librarian . Login via other institutional login options . You can purchase online access to this Article for a 24-hour period (price varies by title) If you already have a Wiley Online Library or Wiley InterScience user account: login above and proceed to purchase the article. If your institution is a registered Wiley Online Library customer, you can log in under your institution's name to see our content. Type your institution's name in the box below. Registered Users please login: Access your saved publications, articles and searches Manage your email alerts, orders and subscriptions Change your contact information, including your password Register now > Local and global motion preferences in descending neurons of the fly. Robust coding of flow-field parameters by axo-axonalgap junctions between fly visual interneurons. Descending neurons. 2009 - Local and global motion preferences in descending neuronsof the fly.
Alexander Borst. Meier M, Serbe E, Maisak MS, Haag J, Dickson BJ, Borst A (2014) Neural circuit components of the Drosophila OFF motion vision pathway.
Curr Biol 24: 385-392. Mauss A, Meier M, Serbe E, Borst A (2014) Optogenetic and pharmacologic dissection of feedforward inhibition in Drosophila motion vision. J Neurosci 34: 2254-2263. Borst A (2014) Neural circuits for elementary motion detection. J Neurogenetics, doi: 10.3109/01677063.2013.876022. Hopp E, Borst A, Haag J (2014) Subcellular mapping of dendritic activity in optic flow processing neurons.
Borst A (2014) Visual motion detection in Drosophila. Cuntz H, Haag J, Borst A (2014) Modelling the cellular mechanisms of fly optic flow processing. Maisak MS, Haag J, Ammer G, Serbe E, Meier M, Leonhardt A, Schilling T, Bahl A, Rubin GM, Nern A, Dickson BJ, Reiff DF, Hopp E, Borst A (2013) A directional tuning map of Drosophila elementary motion detectors. Borst A, Plett J (2013) Seeing the world through an insects eyes. Heller Lecture - Alexander Borst.
In Search of the Holy Grail of Fly Motion Vision.