

jack
I stand for my words
What is Compliance Training? Business Impact With Compliance Training. Compliance training is a significant component for businesses to help companies mitigate and avoid the risk of lawsuits and heavy penalties.
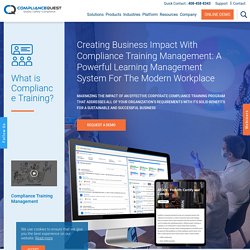
Other enormous benefits of compliance training include:Better productivity in the workplace: Compliance training enables employees to understand how to operate within the law as well as standards that have been introduced by the company to avoid any penalties or lawsuits. Ultimately, when employees are more productive, the company can generate higher profits.Improved workplace culture: Compliance training can help employees understand the rules and regulations so that they can perform their tasks more appropriately and more efficiently. What is CAPA - Corrective and Preventive Action? CAPA Management. Corrective and Preventive Action (CAPA) process helps an organization to analyze and collect relevant product-related information, investigate and identify product and quality problems, and trigger appropriate and effective corrective or preventive actions to eliminate a problem and prevent its recurrence.
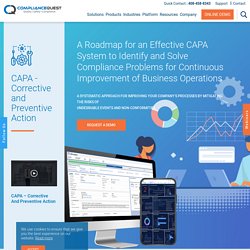
CAPA has two different components, corrective action and preventive action. Corrective action is used to address systemic non-conformities when they occur while preventive actions address the risk of non-conformities that are likely to happen. An efficient and compliant CAPA system needs a set of data in order to identify the problems, implement solutions, and document the outcome and further changes. It is important for employees at all levels, from top management to the floor personnel, to understand the process of CAPA and its importance for an effective system. The initial phase is called identification that collects the information from the event including the following elements: A step-by-step guide for implementing QMS ISO 9001 for Compliance Culture.
A Comprehensive Guide to FDA’s 21 CFR Part 11 Compliance Checklist. An EHS Solution designed to drive an enterprise-wide Safety Culture. A report from McKinsey Global Institute, simply titled “Good health is good business.

Here’s why,” explains the importance of employee health for lasting economic benefits and points out that chronic health issues such as back pain and headache cause a 5% decrease in productivity. Not only is the avoidance of sickness important but also the promotion of employee well-being. Here’s an example of note: A UK-based public sector employer that co-developed and implemented a flexible and multifaceted health promotion toolkit based on an employee survey, was able to reduce absence due to illnesses by 44%, from an average of 4.3 days per employee a year to 2.4 days.
Apart from infections, mental health and ergonomics have also gained importance. The McKinsey report cites the example of a BMW car production plant in Germany that redesigned a production line keeping ergonomics in mind to reduce the physical stress on older workers. ComplianceQuest Solutions. Is Automation Hurting the Health and Safety of the Warehouse Worker? During my career I have worked, walked, and welcomed digitization and automation in the warehouse.

Complete guide on Quality inspection from definition to its implementation. Medical device QMS (Quality Management System) software. What is EHS? Why environmental health and Safety (EHS) is important? Safety management is about preventing incidents proactively.
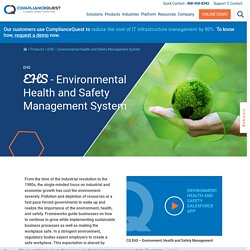
EHS Automation helps organizations stay ahead through a risk-based approach thus improving safety outcomes and promoting organizational and operational excellence. Automatic proactive and reactive EHS reminders are provided to further organizations’ compliance goals in the following ways.Improvement in supplier visibilityIncrease in operating marginA decline in product recalls Safety management is about preventing incidents proactively. Environmental Management Software System. 5 Reasons Why your enterprise must adopt an easily configurable QMS. For a few years now, product management leaders have been “working backwards” to understand “specific” customer needs and building features that “address” those needs.

Late Harvard Business School professor, Clayton Christensen, who is most popular for his pioneering research on disruptive innovation, coined the phrase “Jobs To Be Done” to help innovators understand customer needs at a deeper level. Christensen’s theory is: It is not only about understanding customer needs and wants, but also about what is the end-goal, that “job” a customer needs to get done. EHS and ERP: Do They Go Better Together? Reach Medical Devices Faster to the Customers with FDA’s Emergency Use Authorization (EUA) On March 24, 2020, in response to the insufficient supply of FDA-cleared ventilators for use in healthcare settings to treat patients during the Covid-19 pandemic, the Department of Health and Human Services (HHS) announced Emergency Use Authorization.

Pursuant to section 564(b)(1)(C) of the Federal Food, Drug, and Cosmetic Act (the Act), the EUA authorized the emergency usage of ventilators and the modification of devices such as anesthesia gas machines, positive pressure breathing devices to be used as ventilators or ventilator tubes. This has opened up the possibility of other essential medical devices and products reaching the market faster through the EUA route, especially in emergency situations such as the pandemic. But for that, the products must conform to the criteria for safety, performance, and labeling set forth in Section II and Appendix of the act.
The Guidelines Getting Your Device Added to the EUA List The EUA Process Waivers. New NEP and OSHA Initiative: What Does This Mean For You? Remember back in January when President Biden asked OSHA to do something about COVID-19 and the workforce.

Well, OSHA responded on March 12 of this year with a National Emphasis Program (NEP) with enforcement on companies at which large number of workers face serious risk of COVID-19 infections. The NEP also prioritized whistleblower protection enforcement for workers who face retaliation for reporting unsafe or unhealthy workplace conditions. What Is Quality Audit? Importance of Quality Auditing. What is Quality Assurance? How it is different than Quality Control? What is Cost of Quality? Implementation & Measurement of Cost of Quality. Complete OSHA Inspection checklist and Compliance Checklist. A Complete Guide to OSHA workplace safety standards and regulations. Occupational Safety and Health Act – OSHA. The United States – The OSH Act covers most private sector employers and their workers and some public sector employers and workers in the 50 states and certain territories and jurisdictions under federal authority.
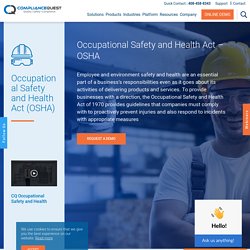
Those jurisdictions include the District of Columbia, Puerto Rico, the Virgin Islands, American Samoa, Guam, Northern Mariana Islands, Wake Island, Johnston Island, and the Outer Continental Shelf Lands as defined in the Outer Continental Shelf Lands Act. Canada – OSHA Compliance and Requirements. OSHA inspectors are called compliance safety and health officers and are experienced, well-trained industrial hygienists and safety professionals. Their role is to assure compliance with OSHA requirements and help employers and workers reduce on-the-job hazards and prevent injuries, illnesses, and deaths in the workplace. Inspections are initiated without advance notice, conducted using on-site or telephone and facsimile investigations, performed by highly trained compliance officers and scheduled based on the following priorities: OSHA inspection officer carries out three important duties during inspection – briefing the employer of the inspection, thorough examination of the site with employee interviews, and summarizing findings in a report.
The first duty of the inspection officer is to brief the employer by in an opening conference where the compliance officer will explain The second duty involves a thorough examination of the site with a walkthrough. OSHA Safety - occupational health and safety administration management system. With Health & safety gaining heightened significance in the current circumstances and with the health & safety heads having to manage remotely, the ComplianceQuest EHS solution enables seamless management of your organization’s complete health and safety management program through a range of intuitively integrated tools and applications.
ComplianceQuest’s Safety, Health, and Environment management system is built from the ground up on the Salesforce platform. This ensures that the EHS is flexible, scalable, and integrates seamlessly with other ERP, CRM, and EQMS platforms. Specifically, it helps build robust processes for the following: Design: Build exhaustive processes, workflows, and checklists to manage workplace safety interventions.Monitor: Track workplace safety data in real-time, with data visibility across the organization.Act: Take action based on predictive analytics and a proactive mindset.Improve: Take corrective or preventive action as needed in a timely manner. Occupational Health and Safety (OHS) Management Systems. Guide to OSHA Compliance, Workplace Safety and Guidelines.
What is EHS? Environment Health and Safety Management – EHS. ComplianceQuest Solutions. Environmental Management Software System. Environment Health Safety and Quality - EHSQ. Most of the time when we talk about EHSQ risk management, we think of health and safety risks.

EHSQ risk management encompasses several types of risk across your enterprise. Below are a few types of EHS risk:Compliance risk: Potential exposure to legal penalties, fines, and losses when a company fails to comply with laws and regulations.Safety risk: A potential exposure to injury, death, or adverse health consequences.Environmental risk: A potential exposure to the negative impact on the environment or living organisms from a company’s emissions, waste, and resource usageClimate risk: Hazards from climate-related events, trends, forecasts and projections.Change Management: Mitigation of risk of injury or accident from inappropriately planned changes in process equipment, chemicals, facilities, etc.
To know how CQ can improve the EHSQ culture in your enterprise, contact us now: