

The Element Sodium. [Click for Isotope Data] Atomic Number: 11 Atomic Weight: 22.98976928 Melting Point: 370.95 K (97.80°C or 208.04°F) Boiling Point: 1156 K (883°C or 1621°F) Density: 0.97 grams per cubic centimeter Phase at Room Temperature: Solid Element Classification: Metal Period Number: 3 Group Number: 1 Group Name: Alkali Metal What's in a name?

Say what? History and Uses: Although sodium is the sixth most abundant element on earth and comprises about 2.6% of the earth's crust, it is a very reactive element and is never found free in nature. Sodium - Periodic Table of Videos. How salt is made - material, used, processing, procedure, industry, machine, Raw Materials, The Manufacturing Process of salt, Quality Control, Health Aspects. Background Salt is the common name for the substance sodium chloride (NaCI), which occurs in the form of transparent cubic crystals.
Although salt is most familiar as a food supplement, less than 5% of the salt produced in the United States is used for that purpose. About 70% is used in the chemical industry, mostly as a source of chlorine. Salt is also used for countless other purposes, such as removing snow and ice from roads, softening water, preserving food, and stabilizing soils for construction. The earliest humans obtained their salt from natural salt concentrations, called licks, and from meat.
As agriculture developed, leading to an increased population and a diet consisting mostly of plants, it became necessary to devise ways of obtaining salt in greater amounts. In areas where the climate did not allow solar evaporation, salt water was poured on burning wood or heated rocks to boil it. Raw Materials.
Sodium Facts - Periodic Table of the Elements Entry for Sodium - Facts about the Element Sodium. Sodium Sodium Basic Facts: Symbol: NaAtomic Number: 11Atomic Weight: 22.989768Element Classification: Alkali MetalCAS Number: 7440-23-5 Sodium Periodic Table Location Group: 1Period: 3Block: s Sodium Electron Configuration Short Form: [Ne]3s1Long Form: 1s22s22p63s1Shell Structure: 2 8 1 Sodium Discovery Discovery Date: 1807Discoverer: Sir Humphrey Davy [England]Name: Sodium derives its name from the Medieval Latin 'sodanum' and the English name 'soda'.
Sodium Physical Data State at room temperature (300 K): SolidAppearance: soft, bright silvery white metalDensity: 0.966 g/ccDensity at Melting Point: 0.927 g/ccSpecific Gravity: 0.971 (20 °C)Melting Point: 370.944 KBoiling Point: 1156.09 KCritical Point: 2573 K at 35 MPa (extrapolated)Heat of Fusion: 2.64 kJ/molHeat of Vaporization: 89.04 kJ/molMolar Heat Capacity: 28.23 J/mol·KSpecific Heat: 0.647 J/g·K (at 20 °C) Sodium Atomic Data Sodium Nuclear Data Number of isotopes: 18 isotopes are known. History of Salt. As far back as 6050 BC, salt has been an important and integral part of the world’s history, as it has been interwoven into the daily lives of countless historic civilizations.
Used as a part of Egyptian religious offerings and valuable trade between the Phoenicians and their Mediterranean empire, salt and history have been inextricably intertwined for millennia, with great importance placed on salt by many different races and cultures of people. Even today, the history of salt touches our daily lives.
The word “salary” was derived from the word “salt.” Salt was highly valued and its production was legally restricted in ancient times, so it was historically used as a method of trade and currency. The word “salad” also originated from “salt,” and began with the early Romans salting their leafy greens and vegetables. Table of Contents: Most people probably think of salt as simply that white granular food seasoning found in a salt shaker on virtually every dining table. Sodium (Na) Saltsense, salt history, salt manufacture, salt uses, sodium. Key information on salt from the Salt Industry.
Salt: Definition, Synonyms from Answers. Because salt is indispensable to life, acts as a food preservative, and uniquely flavors foods, humans have been preoccupied with it since the beginning of recorded history.
The desire to obtain salt politically or militarily has influenced the histories of countries in Asia, Africa, Europe, South America, and the Middle East. Indeed, salt was used as a form of currency and had greater value than gold in some ancient societies. Even religious and magical significance has been attributed to this mineral. In chemistry, the term "salt" generally refers to any compound that results from the interaction of an acid and a base. In the fields of geology and agriculture, the term "salt" is used as a synonym for the word "mineral. " When sodium chloride enters the body, it dissociates almost completely into its constituent particles, the ions sodium and chloride. Sodium Salt Manufacturers,Sodium Salt Manufacturer,Sodium Salt Manufacturers from India. Sodium chloride. Chemistry[edit] Solid sodium chloride[edit] In solid sodium chloride, each ion is surrounded by six ions of the opposite charge as expected on electrostatic grounds.
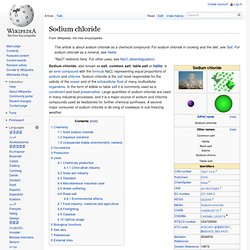
The surrounding ions are located at the vertices of a regular octahedron. In the language of close-packing, the larger chloride ions are arranged in a cubic array whereas the smaller sodium ions fill all the cubic gaps (octahedral voids) between them. This same basic structure is found in many other compounds and is commonly known as the halite or rock-salt crystal structure.
Thermal conductivity of NaCl as a function of temperature has a maximum of 2.03 W/(cm K) at 8 K (−265.15 °C; −445.27 °F) and decreases to 0.069 at 314 K (41 °C; 106 °F). Aqueous solutions[edit] The attraction between the Na+ and Cl− ions in the solid is so strong that only highly polar solvents like water dissolve NaCl well. Unexpected stable stoichiometric variants[edit] Common salt has a well-established 1:1 molar ratio of sodium and chlorine. Uses[edit] Salt. Some of the earliest evidence of salt processing dates back to around 6,000 years ago, when people living in Romania were boiling spring water to extract the salts; a saltworks in China has been found which dates to approximately the same period.
Salt was prized by the ancient Hebrews, the Greeks, the Romans, the Byzantines, the Hittites and the Egyptians. Salt became an important article of trade and was transported by boat across the Mediterranean Sea, along specially built salt roads, and across the Sahara in camel caravans. The scarcity and universal need for salt has led nations to go to war over salt and use it to raise tax revenues.
Salt is also used in religious ceremonies and has other cultural significance. Salt is produced from salt mines or by the evaporation of seawater or mineral-rich spring water in shallow pools.