

Interactions of pH, Carbon Dioxide, Alkalinity and Hardness in Fish Ponds - interactionsphetc.pdf. Inorganic Carbon and pH. Rxn in Aqueous Solution. Common Ion Effect - Equilibrium: Chemistry Tutorial. Kinetics of Minderal Dissolution. Thermodynamics Part 3. Effect of pH on Solubility. Factors that Affect Solubility. The solubility of a substance is affected not only by temperature but also by the presence of other solutes.
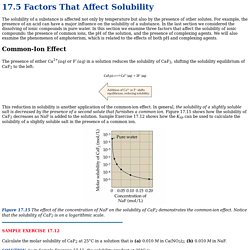
For example, the presence of an acid can have a major influence on the solubility of a substance. In the last section we considered the dissolving of ionic compounds in pure water. In this section we examine three factors that affect the solubility of ionic compounds: the presence of common ions, the pH of the solution, and the presence of complexing agents. We will also examine the phenomenon of amphoterism, which is related to the effects of both pH and complexing agents. Common-Ion Effect. Basic Titrations - Chemistry Tutorial.
Titrate Base With Acid. Also Very Helpful - Carbonate Chemistry. Most Helpful Carbon Dioxide Alkalinity and pH.