

A provider of research peptides.
Backbone-Cyclized Peptides. Backbone cyclization is a technology which is used to make backbone cyclized peptides (BCPs).
There are many chemical methods for implementing this technique, such as the solid-phase coherence strategy, the liquid phase coherence strategy and the natural chemical bonding within molecules and etc. Moreover, BCPs are cyclic peptides formed from the liner peptides through the cyclization between the C-terminal and the N-terminal. It is the cyclic structure between the head and tail makes the BCPs have good enzyme stability, thermal stability and chemical stability.
And, parts of them have membrane permeability. Because of its abnormal stability and high biological activity, it has become a hot spot in the field of drug discovery, as well as the cyclization technology. Application of Backbone-Cyclized Peptides The natural function of the BCPs is host defense, that is, to protect the survival and continuation of this species by inhibiting the growth of alien organisms.
Peptide Hydroxylation. Background Peptide hydroxylation is a post-translational modification catalyzed by 2-oxoglutarate-dependent dioxygenases.
The hydroxylation modification can take place on various amino acids, including but not limited to proline, lysine, asparagine, aspartate and histidine. Hydroxylation of proline or lysine residues in peptides are common post-translational modification event, and such modifications are found in many physiological and pathological processes. Hydroxylation of proline or lysine residues are found in many physiological and pathological processes, and the pattern of these modifications influences many biological functions. Hydroxyproline and hydroxylysine have been found to be implicated in metabolic disorder, connective tissue disorders, lung cancer and stomach cancer. Modification Strategies Proline hydroxylation Lysine hydroxylation The hydroxylation of the lysine residue is catalyzed by lysyl hydroxylase, forming hydroxylysine. Peptide Acetylation.
Acetylation refers to the substitution of hydrogen by acetyl group in organic molecules with oxygen, nitrogen, carbon, sulfur atoms.
Commonly used acylation agents are carboxylic acylation agents, carboxylic ester acylation agents, anhydride acylation agents and acyl chloride acylating agents. Acyl groups are the groups remaining after removal of hydroxyl groups from organic acids, inorganic acids, sulfonic acids, etc. Chemically synthesized peptides often carry free amino groups and free carboxyl groups. The sequence of peptide often represents the sequence of female protein. Peptide Sulfation. Peptide Sulfation is a common post-translational modification of tyrosine residues in eukaryotes that is known to have a widespread occurrence among proteins of multicellular eukaryotic organisms.
Studies on tyrosine-sulfated proteins have shown a major role in enhancing protein-protein interactions, and it is found to be implicated in multiple biological processes. Due to sulfation plays a role in strengthening protein-protein interactions, so types of human proteins known to undergo tyrosine sulfation include G-protein-coupled receptors, adhesion molecules, serine protease inhibitors, coagulation factors, hormones and extracellular matrix proteins. Meanwhile, tyrosine O-sulfate is a stable molecule and is excreted in urine in animals. In addition, studies have shown that it may be observed that tyrosine sulfation has effects on the growth of the mice, such as fecundity, body weight and postnatal viability by knock-out of TPST (tyrosylprotein sulfotransferase) genes in mice.
Peptide Prenylation. Prenylation (also known as isoprenylation or lipidation) is one of the common post-translational peptide modifications, which involves the addition of a farnesyl (C15) or geranylgeranyl (C20) isoprenoid moiety onto a cysteine residue located near the carboxyl terminal of a peptide.
These chemical reactions were catalyzed by farneyl transferase (FTase) and geranylgeranyltransferase (GGTases), respectively. Proteins modified by isoprenylation play an important role in signal transduction in vivo. Fluorescent nanoparticles. Generally speaking, nanoparticles are particles between 1 and 100 nanometres (nm) in diameters with a surrounding interfacial layer contains 20-15000 atoms and are present in a scope between the quantum and newtonian scales. Due to the exceptional characteristic, nanoparticles (NPs) have received much attention in the last decade. Scientific research on nanoparticles is intense as they have many potential applications in medicine, physics, optics, and electronics. For example, NPs can be used to deliver a cargo, such as an anticancer drug, or a cohort of radionuclide atoms to a targeted region of the body, such as a tumor.
Fluorescent Labeling Reagents. Custom Conjugation. * Please be kindly noted that our services can only be used for research to organizations or companies and not intended for individuals. Leveraging our expertise and knowledge in peptides manufacturing, Creative Peptides provides convenient multiple types of peptide conjugation services to meet the needs of different projects, including activating immune responses, obtaining higher stability and cell penetration. We offer peptide conjugation services using antibodies, proteins, oligonucleotides, nanoparticles, etc., as well as other characteristics and quality assessments of the combined products. Tumor Necrosis Factors (TNF) and Related Peptides. Browse products name by alphabetical order: Introduction Tumor Necrosis Factor (TNF) is a kind of cytokine which can kill tumor cells directly without obvious toxicity to normal cells.
All members of TNF family can express transmembrane proteins of type Ⅱ. It consists of three parts: the intracellular region of the amino terminal, the transmembrane region and the extracellular domain of the carboxyl terminal. TNF was mainly produced by activated macrophages, NK cells and T lymphocytes, which were divided into two types: TNF-α and TNF-β.
Vasoactive Intestinal Peptides (VIPs) Browse products name by alphabetical order: Introduction.
Peptide Inhibitors. Antimicrobial Peptides. Browse products name by alphabetical order: Antimicrobial peptides (AMPs), also called host defense peptides (HDPs), which commonly content 5-40 amino acids, are natural antibiotics produced by various organisms.
The characteristics of antimicrobial peptides The first AMP was found by Dubos when extracted an antimicrobial agent from a soil bacillus strain in 1939. This extract was demonstrated to protect mice from pneumococci infection. Since then nearly 5,000 AMPs have been found or synthesized. The largest group corresponds to cationic peptides, which is divided in three classes • The first category composed by linear cationic α-helical peptides, such as Magainin and Cecropins that are linear before their interaction with the cell membrane, and then adopt an amphipathic α-helical secondary structure. Antennapedia Peptides. HIV-TAT Family. A portion of γδT cells bind under MHC-like molecules to identify harmful cells – Creative Peptides Blog. Cells are a key part of our immune system and play a key role in protecting us from harmful pathogens such as viruses and bacteria, as well as cancer.
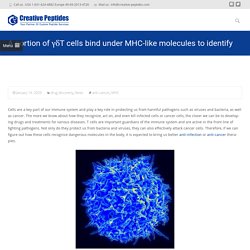
The more we know about how they recognize, act on, and even kill infected cells or cancer cells, the closer we can be to developing drugs and treatments for various diseases. T cells are important guardians of the immune system and are active in the front line of fighting pathogens. Not only do they protect us from bacteria and viruses, they can also effectively attack cancer cells. Therefore, if we can figure out how these cells recognize dangerous molecules in the body, it is expected to bring us better anti-infection or anti-cancer therapies. Figure 1 Scanning electron micrograph of a human T cell In the current mainstream model, T cells identify potential threats through surface “T cell receptors” (TCR). These are important details that can change our approach to studying T cell biology in the future. References Jérôme Le Nours et al. Whether peptide is effective against coronavirus? – Creative Peptides Blog.
The 2019 Novel coronavirus is abbreviated by the World Health Organization.
It is a positive-stranded single-stranded RNA coronavirus with an envelope, which is the pathogen of the pneumonia epidemic caused by the new coronavirus at the end of 2019. Coronavirus is also the “culprit” that caused previous outbreaks of severe acute respiratory syndrome (SARS) and the Middle East respiratory syndrome (MERS). Stapled Peptide Design. Peptide stapling is a strategy for constraining short peptides typically in an alpha-helical conformation. Stapling is carried out by covalently linking the side-chains of two amino acids, thereby forming a peptide macrocycle. Peptide “stapling” via interside-chain hydrocarbon linkages has emerged as one of the most promising approaches for the generation of potent and stable type Ⅰ peptidomimetics of α-helical protein binding motifs.
Once inside the cell, stapled peptides are stable, and able to initiate or inhibit protein-protein interactions that are useful for the treatment of cancer, insulin secretion, inflammation and many other disease states. Chelate Peptides (DOTA related peptides) Peptide-based targeting vectors are becoming increasingly popular for diagnostic and therapeutic applications. They offer several advantages over antibodies including more favorable pharmacokinetics and better tumor penetration. Peptide sequences that are capable of binding to their target with affinities comparable to those of monoclonal antibodies can be generated by chemical or phage display libraries. Affinity selection of random phage display peptide libraries is a very powerful way of identifying novel targeting sequences for a receptor from billions of peptides.
Among many ligands, the macrocyclic 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) stands out, as it forms complexes with Ln(III) ions with extraordinary thermodynamic stability and kinetic inertness. The DOTATOC for scanning DOTA is an organic compound and commonly used as a complexing agent, which has been widely used as contrast agent for cancer treatments. Toxins. Browse products name by alphabetical order: Toxins are poisons produced by organisms.
The term was first coined by the organic chemist Ludwig Brieger. These substances are usually proteins that interfere with other macromolecules in organisms, such as ricin. A substance produced by an organism that can cause animal poisoning in a very small amount. Toxins often differ from other chemicals in their methods of production-the term “toxin” does not specify the method of transport (compared to venom and the wider range of poisons-all substances that may also interfere with organisms).
The seven deadliest chemicals (to humans) are: Five of the seven deadliest compounds known come from nature. Structure Activity Relationship (SAR) Analysis. Overview of SAR Analysis The earliest study of structure-activity relationship (SAR) speculated the relationship between the structure and activity of physiologically active substances, and then inferred the structure of the active site of the target enzyme and designed a new structure of the active substance.
With the development of information technology, at present, the quantitative structure-activity relationship (QSAR) with the computer as an auxiliary tool has become the main direction of structure-activity relationship research. Cosmetic Peptides. Cell Penetrating Peptide Design and Synthesis Services. Disulfide-rich cyclic peptides. Among the variety of post-translational modifications (PTMs), the disulfide bond has gained considerable momentum in biological chemistry as it occurs instantaneously through oxidative folding in peptides, proteins, hormones, enzymes, growth factors, toxins, and immunoglobulins. ACTH and Related Peptide Sequences. Acetalin. Modifiers of Aβ-Aggregation. Melanoma Peptide Sequences. Labeled Amyloid Peptides. Growth Factors and Analogs. Fibronectin Fragments / RGD Peptides. Thymosins and Fragments. Urotensin Related Peptides. APIs. Chimeric Peptides. Nuclear Localization Peptides.
Oligoarginine Peptides. Nucleotides and Nucleosides. Tumor Antigen Derived Peptides. Fluorescence and Dye Labeling Peptides. Photo cross linkers. Peptides-Carraier Protein Conjugation. Peptide Tags. Peptide Target Binder Development. Peptides-Imaging Agents Conjugation. Peptide Mass Fingerprinting. Analytical Method Development and Validation. Post-translational Modification. Custom Peptide Design. De novo Peptide Design.
Peptide Library Design. Recombinant Peptide Synthesis. Oligopeptide Synthesis. Structure Activity Relationship (SAR) Analysis. GMP Grade Peptide Synthesis. Large Scale Peptide Synthesis. Peptide Target Binder Development. Special Antigen Peptides Services. Peptides-Imaging Agents Conjugation. Unusual and Non-natural Amino Acids Modification. MHC-peptides Tetramer Class Ⅰ MHC-peptides Tetramer Class Ⅱ Cyclic Peptides. Peptide PEGylation. C-Terminal Modification-Creative Peptides. Protein Glycosylation. HIV Related Peptides. Peptide N-Terminal Modification. Peptide Modification Services. MHC-Peptide Tetramer. Tripeptide-29. Eye Care. Exendins and Fragments.
Β-Amyloid (1-42), (1-40), (1-46) and Fragments. Insulin-Like Growth Factors (IGF), Fragments & Related Peptides. Chromogranin A / Pancreastatin. Gastric Inhibitory Polypeptide and Fragments. FRET-Substrates. Peptoid Synthesis. Stapled Peptide Synthesis. Long Peptides Synthesis. Peptide Drug Discovery. Peptide Lead Optimization. Acetyl Hexapeptide-38. Acetyl Tetrapeptide-9. Palmitoyl Oligopeptide and Palmitoyl Tetrapeptide-7. Amylins (IAPP) and Fragments.
Peptide drug Bioconjugations Services. Anti-aging. Immunotherapy & Cell Therapy. Peptides in Diabetes. Glycopeptides Synthesis Service. Desmopressin. Surface Plasmon Resonance Imaging (SPRi) Service. Custom Peptide Synthesis. Ghrelin Peptides. Stable Isotope Labeled Peptides. Chelate Peptides (DOTA related peptides) Neoantigen Peptides Vaccine Synthesis Service. Cell Penetrating Peptide Design and Synthesis Services. Matrix Metalloproteinase (MMP) Inhibitors & Substrates.