

Robotics. Robotics is the branch of mechanical engineering, electrical engineering and computer science that deals with the design, construction, operation, and application of robots,[1] as well as computer systems for their control, sensory feedback, and information processing.
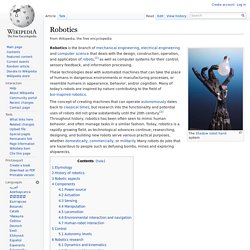
These technologies deal with automated machines that can take the place of humans in dangerous environments or manufacturing processes, or resemble humans in appearance, behavior, and/or cognition. Many of today's robots are inspired by nature contributing to the field of bio-inspired robotics. The concept of creating machines that can operate autonomously dates back to classical times, but research into the functionality and potential uses of robots did not grow substantially until the 20th century.[2] Throughout history, robotics has been often seen to mimic human behavior, and often manage tasks in a similar fashion. Etymology[edit] History of robotics[edit] Robotic aspects[edit] Components[edit] Power source[edit]
Electron deficiency. Electron deficiency occurs when a compound has too few valence electrons for the connections between atoms to be described as covalent bonds.
Electron deficient bonds are often better described as 3-center-2-electron bonds. Examples of compounds that are electron deficient are the boranes. The term electron-deficient is also used in a more general way in organic chemistry, to indicate a pi-system such as an alkene or arene that has electron-withdrawing groups attached, as found in nitrobenzene or acrylonitrile. Instead of showing the nucleated character common with simple C=C bonds, electron-deficient pi-systems may be electrophilic and susceptible to nucleophilic attack, as is seen in the Michael addition or in nucleophilic aromatic substitution. Nuclide. A nuclide (from nucleus) is an atomic species characterized by the specific constitution of its nucleus, i.e., by its number of protons Z, its number of neutrons N, and its nuclear energy state.[1] The word nuclide was proposed [2] by Truman P.
Kohman [3] in 1947. Matter. Before the 20th century, the term matter included ordinary matter composed of atoms and excluded other energy phenomena such as light or sound.
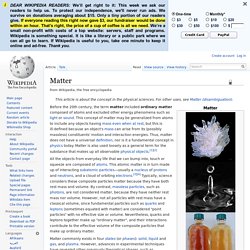
This concept of matter may be generalized from atoms to include any objects having mass even when at rest, but this is ill-defined because an object's mass can arise from its (possibly massless) constituents' motion and interaction energies. Thus, matter does not have a universal definition, nor is it a fundamental concept in physics today. Matter is also used loosely as a general term for the substance that makes up all observable physical objects.[1][2] All the objects from everyday life that we can bump into, touch or squeeze are composed of atoms.
This atomic matter is in turn made up of interacting subatomic particles—usually a nucleus of protons and neutrons, and a cloud of orbiting electrons.[3][4] Typically, science considers these composite particles matter because they have both rest mass and volume. Definition Common definition Quarks. Isotope. The three naturally-occurring isotopes of hydrogen.
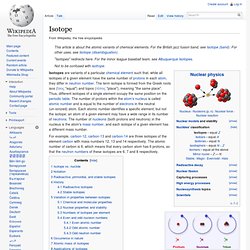
The fact that each isotope has one proton makes them all variants of hydrogen: the identity of the isotope is given by the number of neutrons. From left to right, the isotopes are protium (1H) with zero neutrons, deuterium (2H) with one neutron, and tritium (3H) with two neutrons. Isotopes are variants of a particular chemical element such that, while all isotopes of a given element have the same number of protons in each atom, they differ in neutron number. The term isotope is formed from the Greek roots isos (ἴσος "equal") and topos (τόπος "place"), meaning "the same place". Neutron number. This diagram shows the half-life (T½) of various isotopes with Z protons and neutron number N.
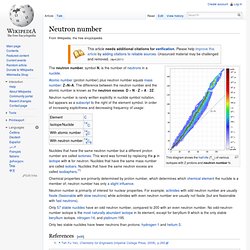
The neutron number, symbol N, is the number of neutrons in a nuclide. Atomic number. Molecule. Lorentz force. In physics, particularly electromagnetism, the Lorentz force is the combination of electric and magnetic force on a point charge due to electromagnetic fields.
If a particle of charge q moves with velocity v in the presence of an electric field E and a magnetic field B, then it will experience a force. For any produced force there will be an opposite reactive force. Neutron. The neutron is a subatomic hadron particle that has the symbol n or n0.
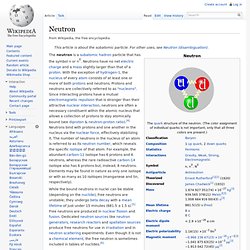
Neutrons have no net electric charge and a mass slightly larger than that of a proton. With the exception of hydrogen-1, the nucleus of every atom consists of at least one or more of both protons and neutrons. Protons and neutrons are collectively referred to as "nucleons". Since interacting protons have a mutual electromagnetic repulsion that is stronger than their attractive nuclear interaction, neutrons are often a necessary constituent within the atomic nucleus that allows a collection of protons to stay atomically bound (see diproton & neutron-proton ratio).[4] Neutrons bind with protons and one another in the nucleus via the nuclear force, effectively stabilizing it. Proton. Electron. History[edit] In the early 1700s, Francis Hauksbee and French chemist Charles François de Fay independently discovered what they believed were two kinds of frictional electricity—one generated from rubbing glass, the other from rubbing resin.
From this, Du Fay theorized that electricity consists of two electrical fluids, vitreous and resinous, that are separated by friction, and that neutralize each other when combined.[17] A decade later Benjamin Franklin proposed that electricity was not from different types of electrical fluid, but the same electrical fluid under different pressures. Atomic nucleus. A model of the atomic nucleus showing it as a compact bundle of the two types of nucleons: protons (red) and neutrons (blue).
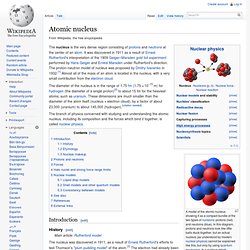
In this diagram, protons and neutrons look like little balls stuck together, but an actual nucleus (as understood by modern nuclear physics) cannot be explained like this, but only by using quantum mechanics. In a nucleus which occupies a certain energy level (for example, the ground state), each nucleon has multiple locations at once. Atom. Entropy (order and disorder) Boltzmann's molecules (1896) shown at a "rest position" in a solid In thermodynamics, entropy is commonly associated with the amount of order, disorder, and/or chaos in a thermodynamic system. This stems from Rudolf Clausius' 1862 assertion that any thermodynamic processes always "admits to being reduced to the alteration in some way or another of the arrangement of the constituent parts of the working body" and that internal work associated with these alterations is quantified energetically by a measure of "entropy" change, according to the following differential expression:[1] In the years to follow, Ludwig Boltzmann translated these "alterations" into that of a probabilistic view of order and disorder in gas phase molecular systems.
Locally, the entropy can be lowered by external action. Entropy. Where T is the absolute temperature of the system, dividing an incremental reversible transfer of heat into that system (dQ). (If heat is transferred out the sign would be reversed giving a decrease in entropy of the system.) The above definition is sometimes called the macroscopic definition of entropy because it can be used without regard to any microscopic description of the contents of a system. The concept of entropy has been found to be generally useful and has several other formulations.