

Periodic table. Standard 18-column form of the periodic table. The colors used in the version of the table shown here signify different categories of elements, as listed below in the Layout section, under the larger table. The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, any such table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table—whether in the standard form or some other variant—provides a useful framework for analyzing chemical behavior, and such tables are widely used in chemistry and other sciences.
Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. Layout Grouping methods Groups Periods Blocks Categories Atomic radii. Chemical element. Chemical bond. A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds.
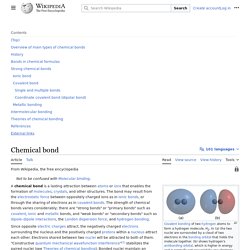
The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds; or through the sharing of electrons as in covalent bonds. The strength of chemical bonds varies considerably; there are "strong bonds" or "primary bonds" such as covalent or ionic or metallic bonds, and "weak bonds" or "secondary bonds" such as dipole–dipole interactions, the London dispersion force and hydrogen bonding. Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position.
This attraction constitutes the chemical bond. Chemical substance. Steam and liquid water are two different forms of the same chemical substance, water.
In chemistry, a chemical substance is a form of matter that has constant chemical composition and characteristic properties.[1] It cannot be separated into components by physical separation methods, i.e. without breaking chemical bonds. It can be solid, liquid, gas, or plasma. Forms of energy, such as light and heat, are not considered to be matter, and thus they are not "substances" in this regard. Definition[edit] Colors of a single chemical (Nile red) in different solvents, under visible and UV light. Chemical substances (also called pure substances) may well be defined as "any material with a definite chemical composition" in an introductory general chemistry textbook.[2] According to this definition a chemical substance can either be a pure chemical element or a pure chemical compound.
History[edit] Chemical elements[edit] Chemical compounds[edit] Substances versus mixtures[edit] Naming and indexing[edit] Chemical reaction. A thermite reaction using iron(III) oxide.
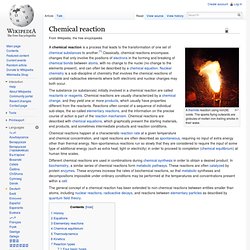
The sparks flying outwards are globules of molten iron trailing smoke in their wake. A chemical reaction is a process that leads to the transformation of one set of chemical substances to another.[1] Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive elements where both electronic and nuclear changes may both occur. Chemical reactions happen at a characteristic reaction rate at a given temperature and chemical concentration, and rapid reactions are often described as spontaneous, requiring no input of extra energy other than thermal energy.
History Equations Dissociation of a molecule AB into fragments A and B. Chemistry. Chemistry, a branch of physical science, is the study of the composition, structure, properties and change of matter.[1][2] Chemistry is chiefly concerned with atoms and their interactions with other atoms - for example, the properties of the chemical bonds formed between atoms to create chemical compounds.
As well as this, interactions including atoms and other phenomena - electrons and various forms of energy—are considered, such as photochemical reactions, oxidation-reduction reactions, changes in phases of matter, and separation of mixtures. Finally, properties of matter such as alloys or polymers are considered. Chemistry is sometimes called "the central science" because it bridges other natural sciences like physics, geology and biology with each other.[3][4] Chemistry is a branch of physical science but distinct from physics.[5] Etymology The word alchemy in turn is derived from the Arabic word al-kīmīā (الکیمیاء). Definition History.